Clinical Characteristics of Glaucomatous Subjects Treated with Refractive Corneal Ablation Surgery
Article information
Abstract
Purpose
To evaluate the clinical characteristics of newly diagnosed glaucomatous subjects who had a history of refractive corneal ablation surgery (RCAS).
Methods
Sixty-eight glaucomatous subjects who had a history of RCAS and 68 age- and visual field (VF) mean deviation-matched glaucomatous subjects with no history of RCAS were included. Intraocular pressure (IOP), central corneal thickness (CCT), VF, and retinal nerve fiber layer thickness determined by optical coherence tomography were assessed. Parameters were compared between patients with and without a history of RCAS. Between-eye comparisons in the same participant (more advanced vs. less-advanced eye, in terms of glaucoma severity) were performed in the RCAS group.
Results
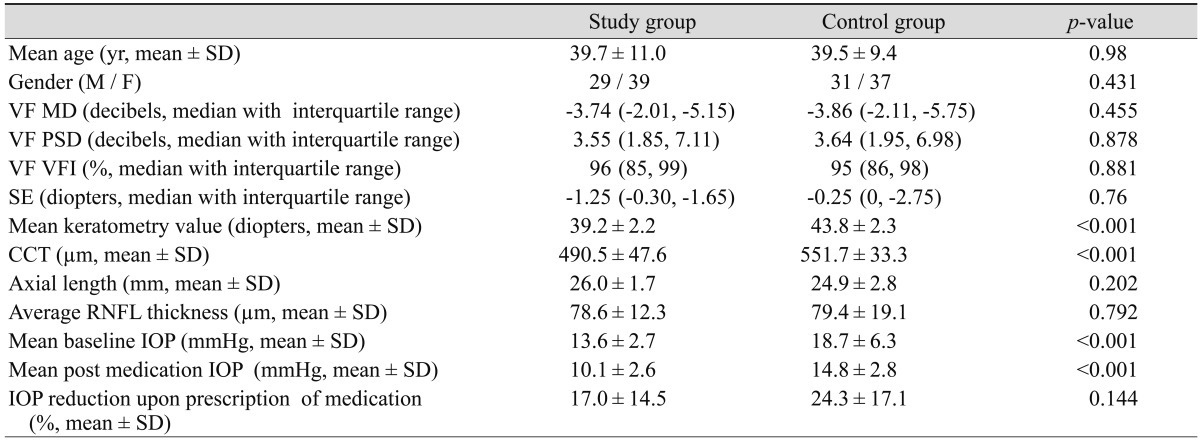
With similar levels of glaucoma severity, those with a history of RCAS showed significantly lower baseline IOP and a thinner CCT than the eyes of individuals without a RCAS history (13.6 vs. 18.7 mmHg, 490.5 vs. 551.7 µm, all p < 0.001). However, the extent of IOP reduction after anti-glaucoma medication did not significantly differ between the two groups (17% vs. 24.3%, p = 0.144). In the between-eye comparisons of individual participants in the RCAS group, the more advanced eyes were more myopic than the less-advanced eyes (-1.84 vs. -0.58 diopter, p = 0.003).
Conclusions
Eyes with a history of RCAS showed a similar level of IOP reduction as eyes without such a history after anti-glaucoma medication. Our finding that the more advanced eyes were more myopic than the less-advanced eyes in the same participant may suggest an association between glaucoma severity and myopic regression.
Refractive corneal ablation surgery (RCAS) using a laser, such as photorefractive keratectomy (PRK), laser in situ keratomileusis (LASIK), or laser in situ epithelial keratomileusis (LASEK), has become popular for the treatment of refractive errors. Because most RCAS candidates are myopes and because glaucoma is more prevalent in such subjects, the safety of RCAS in terms of glaucoma development or progression is frequently questioned by those who wish to undergo such surgery. One concern is related to the reliability of intraocular pressure (IOP) measurements obtained after surgery. As most current IOP measurement procedures are performed through the cornea, corneal ablation inevitably affects IOP readings [1-8]. Inaccurate measurements of IOP, and the subsequent possibility that IOP measurements cannot be used as a reliable guideline for the diagnosis and management of glaucoma, have become issues warranting attention.
Another concern is whether the surgical process or the post-operative management may cause glaucomatous damage to develop or become aggravated. For example, the possibility of optic nerve damage during acute IOP elevation has been suggested to occur when the suction ring of a microkeratome is placed [9]. Postoperative IOP elevation caused by steroid use has also been reviewed [10-13]. However, we could not find reports on the characteristics of glaucoma patients with a history of RCAS. Therefore, we examined the characteristics of such patients.
Materials and Methods
Subjects
Subjects examined at the glaucoma clinic of the Asan Medical Center, Seoul, Korea, from March 2009 to January 2011 and who met the inclusion criteria described below were retrospectively selected for medical record review. Inclusion criteria consisted of a history of bilateral uneventful RCAS for the treatment of myopia and a diagnosis of glaucoma in at least one eye for the first time. Therefore, no participant had a history of glaucoma prior to RCAS. To allow for between-eye comparisons, we included subjects who had undergone bilateral RCAS. The control group consisted of age- and glaucoma severity (visual field [VF] mean deviation [MD])-matched subjects who had no history of RCAS and who were newly diagnosed with glaucoma.
At the time of initial diagnosis, each patient received a comprehensive ophthalmologic examination including the following components: a review of medical history; measurement of best-corrected visual acuity (BCVA) to confirm that visual acuity was adequate for the performance of automated perimetry, slit-lamp biomicroscopy, autorefractometry, autokeratometry, axial length measurement by IOL master (Carl Zeiss Meditec, Dublin, CA, USA), IOP measurement by Goldmann applanation tonometry (GAT), gonioscopy, a dilated fundoscopic examination using a 90- or 78-diopter (D) lens, stereoscopic optic disc photography, repeated VF examination by standard automated perimetry (Swedish interactive threshold algorithm standard strategy 24-2, Carl Zeiss Meditec), central corneal thickness (CCT; DGH-550 instrument, DGH Technology Inc., Exton, PA, USA) assessment, and optical coherence tomography (OCT) retinal nerve fiber layer (RNFL) thickness measurement (Cirrus HD OCT; Carl Zeiss Meditec).
For inclusion in the study, all patients needed to have at baseline examination a BCVA of 20 / 30 or better and the presence of a normal anterior chamber and open-angle on slit-lamp and gonioscopic examinations. Patients with concomitant diseases known to affect the VF, other than glaucoma, were excluded.
The diagnosis of glaucoma was based on the presence of typical glaucomatous optic disc changes (disc excavation, diffuse or focal neural rim thinning, disc hemorrhage, or RNFL defects) and glaucomatous VF defects agreed upon by two glaucoma experts (JHN and KRS). Eyes with glaucomatous VF defects were defined as those that met at least two of the following criteria, as confirmed by more than two reliable consecutive test results, in addition to compatibility with optic nerve appearance: 1) a cluster of three points with a probability of less than 5% on a pattern deviation map in at least one hemifield, including at least one point with a probability of less than 1% or a cluster of two points with a probability of less than 1%; 2) a glaucoma hemifield test result outside normal limits; and/or 3) a pattern standard deviation (PSD) outside 95% of the normal limits. Reliable VF assessment was defined as a VF test with a false-positive error <15%, a false-negative error <15%, and a fixation loss <20%. To minimize any learning effect, the first VF test was excluded from analysis.
All procedures conformed to the Declaration of Helsinki and the study was approved by the institutional review board of the Asan Medical Center at the University of Ulsan, Seoul, Korea.
Analysis
The Wilk-Shapiro test was used to explore the distribution of numerical data. Normally distributed data are presented as means with standard deviations, whereas non-normally distributed data are shown as medians with interquartile ranges. Normally distributed data were compared between study (glaucomatous eyes with a history of RCAS) and control eyes (glaucomatous eyes with no history of RCAS) using the unpaired t-test. Non-normally distributed data were compared employing the Mann-Whitney test. To compare categorical data, the chi-square test was used. In comparisons between the study and control groups, one eye was randomly selected if both eyes were eligible. Spherical equivalent (SE) values determined by autorefractometry, mean keratometry values, mean baseline IOPs, IOP at 1 month post-IOP lowering treatment, axial length, CCT, and RNFL thickness as determined by OCT were compared between the study and control group.
Between-eye comparisons ('better eye' vs. 'worse eye' in the same subject of the RCAS group) were performed using the paired t-test (normally distributed data) or the Wilcoxon signed rank test (non-normally distributed data). If only one eye had glaucoma, this eye was considered to be the 'worse eye.' If both eyes had glaucoma, the more advanced eye in terms of glaucoma severity, as assessed by VF MD, was categorized as the 'worse eye.' All of the measurements of SE, IOP, CCT, axial length, and RNFL thickness as determined by OCT were compared between the better and the worse eyes.
All p-values less than 0.05 were considered statistically significant. SPSS ver. 17.0 (SPSS Inc., Chicago, IL, USA) and MedCalc ver. 9.6 (MedCalc, Mariakerke, Belgium) were used for statistical analysis.
Results
Sixty-eight Korean glaucomatous subjects with a history of RCAS constituted the study group. Twenty nine subjects were men and 39 were women. Eight subjects had undergone PRK, 47 had undergone LASIK, and 13 had undergone LASEK. The time that had elapsed since RCAS ranged from 1 to 16 years (median, 6 years). No subject in the study group had ever been diagnosed with glaucoma. All subjects indicated that the RCAS treatment had fully corrected their myopia. No subject experienced IOP elevation after RCAS for any other reason. Forty-nine subjects were referred from general health care centers because of optic disc features that had raised the suspicion of glaucoma, whereas 19 were referred from primary-care eye clinics for the same reason.
Mean age, VF MD, PSD, and VF index did not differ significantly between the study and control group subjects because we matched them in terms of age and glaucoma severity (p = 0.98, 0.455, 0.878, and 0.881, respectively). The SE, axial length, and RNFL thickness values did not differ between the two groups (p = 0.76, 0.202, and 0.792, respectively). However, in the study group, mean baseline IOP was significantly lower (13.6 vs. 18.7 mmHg, p < 0.001), CCT was thinner (490.5 vs. 551.7 µm, p < 0.001), and the mean keratometry value was lower (39.2 vs. 43.8 D, p < 0.001). Fifty-seven eyes of the study group (84%) and 60 of the control group (88%) underwent treatment with anti-glaucoma medication. Mean IOP at 1 month post-IOP lowering treatment was significantly lower in the study group (10.1 vs. 14.8 mmHg, p < 0.001); however, the extent of IOP reduction did not significantly differ between the two groups (17.0% vs. 24.3%, p = 0.144) (Table 1).
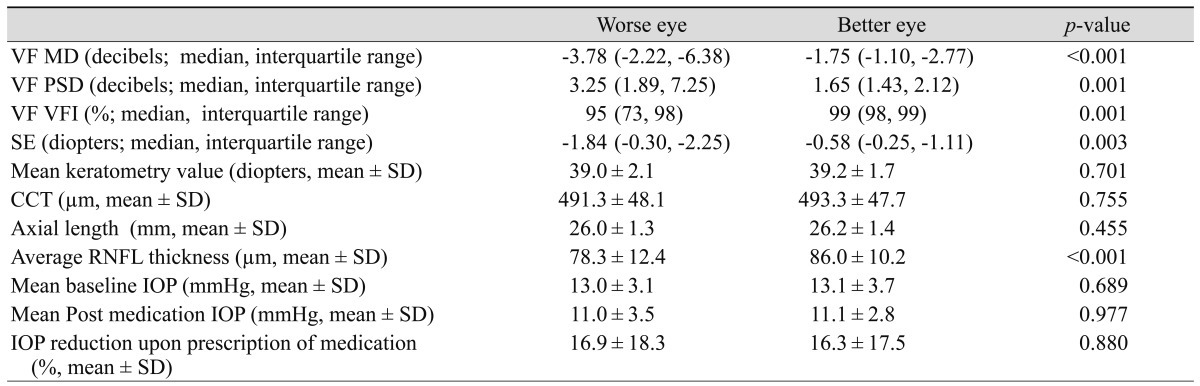
When between-eye comparisons were conducted in the RCAS group, the median (interquartile range) VF MD was -3.78 dB (-2.22, -6.38) in the worse eye and -1.75 dB (-1.10, -2.77) in the better eye. The worse eye was significantly more myopic than the better eye (median [interquartile range], -1.84 D [-0.30, -2.25] vs. -0.58 D [-0.25, -1.11]; p = 0.003), [interquartile range], -1.84 D. However, other factors, including mean baseline and post-medication IOP, axial length, mean keratometry value, and CCT did not differ significantly between the better and worse eyes (Table 2).
Discussion
RCAS has become very popular worldwide for the treatment of refractive errors, especially myopia. An increasing number of myopic subjects are expected to receive RCAS in the future. Glaucoma is sometimes diagnosed in RCAS candidates upon preoperative examination and is also seen in some subjects with a history of RCAS. Although concerns have been raised about possible inconsistencies in IOP measurements conducted before and after RCAS and IOP elevation upon steroid use after the procedure, the clinical characteristics of glaucoma patients who have undergone RCAS have not been studied [1-8,10-13].
In the present work, CCT, mean keratometry values, and both baseline and post-medication IOPs were significantly lower in eyes with a history of RCAS. As all IOP measurements were performed using GAT, a lower IOP reading can be attributed to the presence of a thinner and flatter cornea after RCAS. Lower IOP GAT readings after RCAS have been reported in previous studies [1-8]. Considering that the subjects in this study underwent RCAS up to 16 years ago (with a median time of 6 years), it is clear that the lower IOP readings are not a temporary phenomenon confined to a limited period after the operation. RCAS reduces corneal thickness and flattens corneal curvature, and GAT measures the pressure needed for the flattening of the cornea. Hence, both flattening and thinning of the cornea appear to affect the IOP readings taken by GAT. We must emphasize the importance of preoperative and follow-up documentation of optic disc and VF in subjects scheduled for RCAS. We found that glaucomatous eyes in the RCAS group showed a significantly lower IOP as measured by GAT, approximately 6 mmHg below that of the control group. In fact, no RCAS group member was referred to us because of IOP elevation. This suggests that the presence of glaucoma may be difficult to detect by IOP measurement in an eye with a history of RCAS. Careful optic disc examinations and subsequent VF testing if indicated are crucial for the early diagnosis of glaucoma in patients with a history of RCAS.
Interestingly, although both pre- and post-medication IOPs were lower in the RCAS subjects than the controls, the extent of IOP reduction did not significantly differ between the two groups. In other words, a similar level of IOP reduction was achieved in both groups after prescription of anti-glaucoma medication. These observations suggest that IOP measurement by GAT can still be used as a guideline for follow-up IOP measurements in glaucomatous subjects, despite the lower IOP readings obtained using GAT after RCAS.
Glaucoma usually presents asymmetrically. Therefore, a cup-to-disc ratio that diverges from unity is considered to be an early sign of glaucomatous optic disc change. In subjects with a history of RCAS, we wished to determine which eye was more vulnerable to glaucoma development or progression by assessing the differences in the clinical characteristics between the more advanced and less advanced eye in terms of glaucoma severity within the same participant. Interestingly, the more advanced eye was found to be more myopic. This may suggest a relationship between myopic regression and glaucoma development or progression. The eye in which myopia tends to redevelop more easily may be more vulnerable to glaucomatous damage. Previous reports have suggested that a higher IOP may contribute to myopic regression by shifting the cornea forward [14-16]. Thus, some clinicians have suggested the use of IOP-lowering medication to halt myopic regression and have reported favorable outcomes [14,15,17]. We speculate that although IOP readings are artificially lower after RCAS, a relatively higher genuine IOP induced both myopic shifts and glaucomatous optic disc and VF damage. However, our present study was cross-sectional in design, not prospective. Thus, pre- and post-RCAS medical records were not available. Based on self-reports, we assumed that all eyes were not glaucomatous before RCAS, were treated to induce full correction, and were all emmetropic immediately after RCAS. Predictive risk factors for myopic regression after RCAS include a greater ablation depth, lower preoperative CCT, higher IOP, greater extent of attempted correction, and higher preoperative myopic degree [14,16,18]. The observation that the axial length of the two groups was not significantly different may suggest that the preoperative myopic degree in both groups was not different as well. However, as peri-RCAS information was not available for our study participants, we could not investigate the effects of such factors on myopic regression and glaucoma status. Therefore, our observations in this regard should be interpreted with caution.
Prospective evaluations of the effect of RCAS on patients with established glaucoma may be difficult to perform because conducting RCAS on eyes with a proven pathology such as glaucoma may not be acceptable in many circumstances. Hence, it may be difficult to study the influence of RCAS on the development or progression of glaucoma using prospective long-term follow-up studies.
Although we cannot prove a relationship between myopic regression and vulnerability to glaucoma because of inherent limitations of our study design, the issue warrants exploration. Myopic regression after RCAS is known to be related to a higher level of myopia, a thinner CCT, and elevated IOP [14,16,18]. These are also risk factors for glaucoma development and progression [19-23]. The lamina cribrosa (LC) has been suggested as the principal focus of glaucoma development. However, at the present time, the LC cannot be studied in great detail. Thus, corneal properties are used as surrogate measures providing information on LC status, as these two tissues share an embryological origin [24-26]. A study of the effect of corneal ablation on LC would be interesting and could provide the theoretical background necessary for work exploring the influence of RCAS on glaucoma.
Finally, we believe that the most important issue is the clinical course of glaucoma patients with a history of RCAS. Based on our current cross-sectional report, further data on longitudinal IOP measurements, the response of IOP to anti-glaucoma medication, optic disc and VF changes after the use of various treatments, and comparisons between glaucoma patients with and without a history of RCAS should be collected in a forthcoming study.
In conclusion, when eyes showing similar levels of glaucoma severity were examined, eyes with a history of RCAS showed significantly lower IOP readings than did those without such a history. However, eyes with and without a history of RCAS showed similar levels of IOP reduction after the prescription of anti-glaucoma medication. Thus, the measurement of IOP remains important during follow-up. When both eyes of RCAS group subjects were examined, the more advanced glaucomatous eye tended to be more myopic than the less advanced eye, which may suggest an association between glaucoma development and myopic regression. We believe that this issue warrants further investigation.
Notes
No potential conflict of interest relevant to this article was reported.