Incidence of Acute Endophthalmitis after Intravitreal Antivascular Endothelial Growth Factor Injection in Age-related Macular Degeneration
Article information
Abstract
Purpose
Antivascular endothelial growth factor (anti-VEGF) agents are routinely intravitreously injected to treat neovascular age-related macular degeneration (AMD). Postoperative endophthalmitis, a side effect after intravitreal anti-VEGF injection, has been reported to have a low incidence but may threaten vision. In this study, we aimed to analyze the incidence of acute endophthalmitis after intravitreal anti-VEGF injection and associated risk factors in patients with neovascular AMD in South Korea.
Methods
Using the health claims data recorded in the Korean National Health Insurance System database, we identified newly developed neovascular AMD cases from 2010 through 2019. Acute endophthalmitis was defined as a case of invasive treatment for accompanying symptoms. All statistical analyses were performed with a significance level p < 0.05. To find risk factors, we used univariate and multivariable Poisson regression.
Results
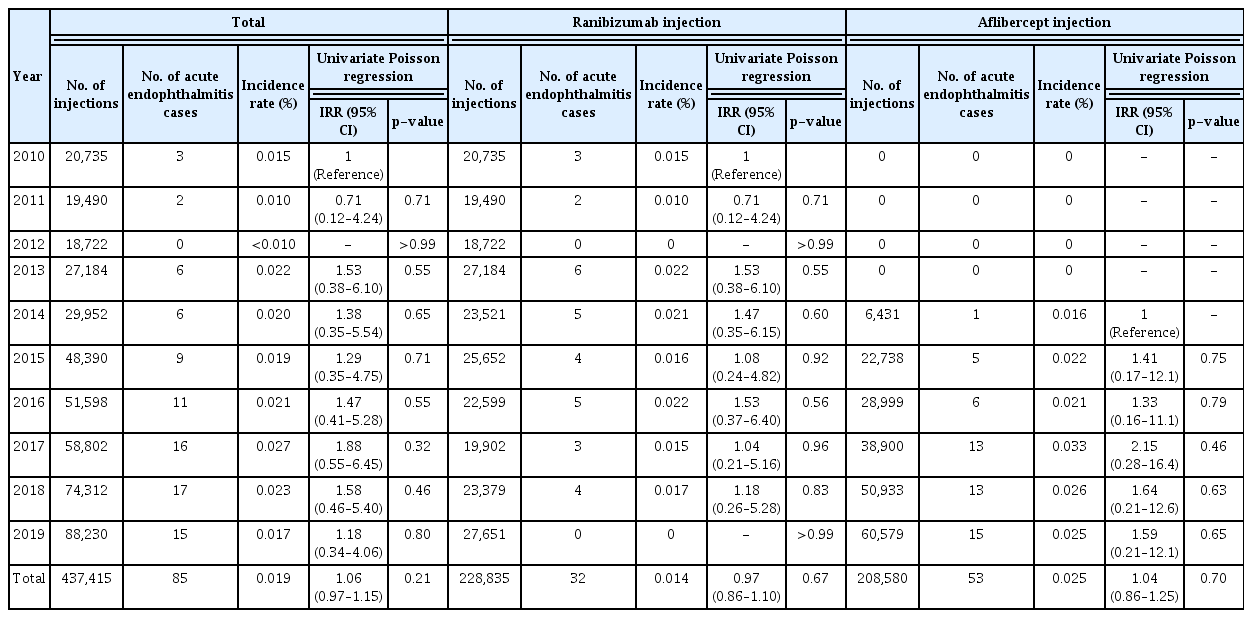
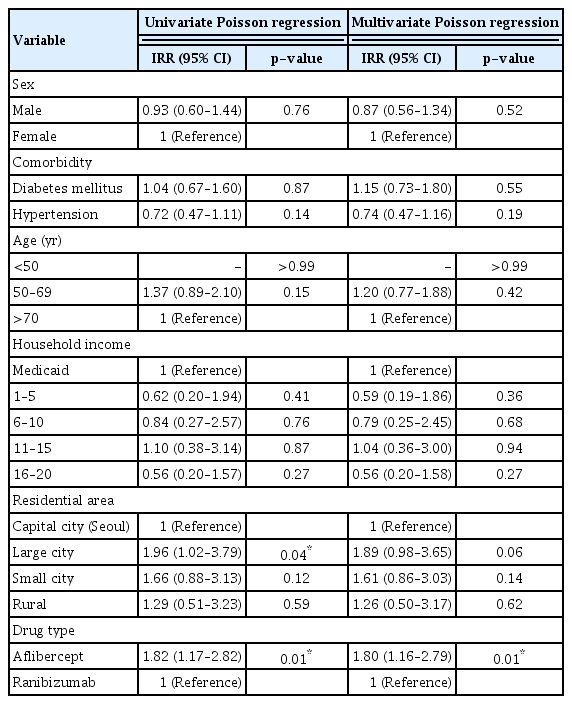
The overall incidence of acute endophthalmitis was 0.019% (p = 0.21) during the 10-year period: 0.025% for aflibercept injection and 0.014% for ranibizumab injection. The incidence was higher in metropolitan city residents than in Seoul (incidence rate ratio [IRR], 1.96; 95% confidence interval [CI], 1.02–3.79; p = 0.04) and was higher for aflibercept injections than for ranibizumab injections (IRR, 1.82; 95% CI, 1.17–2.82; p = 0.01). However, in multivariate analysis, only aflibercept injections showed a significant effect on the incidence of acute endophthalmitis (IRR, 1.80; 95% CI, 1.16–2.79; p = 0.01).
Conclusions
The incidence of acute endophthalmitis after intravitreal anti-VEGF injections was generally low, and aflibercept was revealed as a significant risk factor.
Globally, age-related macular degeneration (AMD) is one of the leading causes of visual impairment and blindness in people aged >60 years in developed countries [1]. The prevalence of AMD is gradually increasing owing to an increase in the population of older adults [2]. Of the two types of AMD, atrophic or neovascular, the neovascular type is characterized by the presence of new abnormal blood vessels in the subretinal space, resulting in more severe vision loss.
Antivascular endothelial growth factor (anti-VEGF) agents have been widely used to treat neovascular AMD. The most commonly used anti-VEGF agents are aflibercept (Eylea; Regeneron, Tarrytown, NY, USA), ranibizumab (Lucentis; Genentech, South San Francisco, CA, USA), and bevacizumab (Avastin, Genentech). Both aflibercept and ranibizumab are supplied in single-use vials. In case of prefilled formulation, aflibercept is not currently available on the market in South Korea, while ranibizumab has been approved since June 2019. Meanwhile, bevacizumab, an off-label drug, is used without regulatory approval and is not listed by the Korean National Health Insurance System (KNHIS) for neovascular AMD [3].
Acute endophthalmitis is a sight-threatening complication of intravitreal anti-VEGF injections [4]. The reported incidence of endophthalmitis following intravitreal anti-VEGF injection is generally low, ranging from 0.01% to 0.44% [5–11]. Endophthalmitis can cause considerable vision loss, and a thorough understanding of its incidence and risk factors is important. However, to date, no previous study in South Korea has analyzed the incidence of acute endophthalmitis after intravitreal injection in neovascular AMD by using entire population data, while only a few such studies have been conducted worldwide. Therefore, this study aimed to analyze the incidence of acute endophthalmitis after intravitreal anti-VEGF injection and the associated risk factors in patients with neovascular AMD based on claims data from the KNHIS database.
Materials and Methods
Ethics statement
This retrospective, nationwide, population-based study was approved by the Institutional Review Board of the National Health Insurance Service Ilsan Hospital (No. NHIMC 2020-12-019). The study adhered to the tenets of the Declaration of Helsinki, and the requirement for written informed consent was waived because all data provided by the KNHIS were anonymized.
Data source
We accessed the health claims data recorded in the KNHIS database. In South Korea, approximately 97% of all Korean nationals are registered under the KNHIS, and the remaining 3% are covered by the Medical Assistance Program. The KNHIS database includes information pertaining to patients’ sociodemographic characteristics, diagnostic codes, procedures, prescription drugs, and direct medical costs for both inpatient and outpatient care.
Data extraction
The Korean Ministry of Health and Welfare classified neovascular AMD as a “rare disease” in August 2009, and patients with the neovascular AMD registration code (V201) were entitled to receive benefits from a copayment assistance policy from August 2009 forward. We identified newly developed neovascular AMD cases using the registration code V201 from January 1, 2010, through December 31, 2019. Patients who were prescribed ranibizumab and aflibercept were included. Patients younger than 40 years of age or those without data for demographic variables were excluded. We collected data on sex, age, comorbidities, household income, residential area, number of intravitreal injections, and type of anti-VEGF medication; aflibercept supplied in single-use vials and ranibizumab supplied in both vial and prefilled syringe.
We assumed that if endophthalmitis occurred within 4 weeks after intravitreal anti-VEGF injection, it was acute endophthalmitis caused by the injection. Acute endophthalmitis was defined as cases in which treatment with vitrectomy (Korean Electronic Data Interchange [KEDI] codes S5121, S5122) and intravitreal antibiotic injection (KEDI code S5070) were performed on the same day or intravitreal antibiotic injection alone was performed under the diagnosis of acute endophthalmitis (Korean Standard Classification of Disease-8 [KCD-8] codes H44.0, H44.1, and H45.1). Recurrences within 4 weeks were excluded.
Statistical analysis
The incidence of acute endophthalmitis per injection was calculated and analyzed for differences in annual incidence over time. Using univariate and multivariable Poisson regression, we also evaluated the relationship between the onset of endophthalmitis and sex, age, systemic diseases, household income, residential area, and type of medication. The significance level was set at p < 0.05. All statistical analyses were performed using the SAS ver. 9.4 (SAS Institute Inc., Cary, NC, USA).
Results
Incidence of acute endophthalmitis
From 2010 to 2019, a total of 437,415 intravitreal anti-VEGF injections were administered (ranibizumab, 52.3%; aflibercept, 47.7%), and acute endophthalmitis was diagnosed in 85 patients. Table 1 shows the distribution of intravitreal injections from 2010 to 2019 for the entire country as well as the distribution for each medication. The overall incidence of acute endophthalmitis was 0.019% (p = 0.21) during this 10-year period, and there was no significant difference in incidence by year. The incidence was higher after aflibercept injection (0.025%) than after ranibizumab injection (0.014%) (Table 1).
Period from injection to acute endophthalmitis
Acute endophthalmitis occurred within 1 week after intravitreal injection in 65 cases (76.5%) and within 2 weeks in 13 cases (15.3%). Subsequently, three and four cases of endophthalmitis occurred at the 3rd and 4th weeks, respectively (Table 2).
Risk factors for acute endophthalmitis
We analyzed the risk factors associated with the onset of acute endophthalmitis after intravitreal anti-VEGF injections. In univariate analysis, the incidence was higher in metropolitan city residents than in capital city Seoul residents (incidence rate ratio [IRR], 1.96; 95% confidence interval [CI], 1.02–3.79; p = 0.04) and was higher for aflibercept injections than for ranibizumab injections (IRR, 1.82; 95% CI, 1.17–2.82; p = 0.01). In multivariate analysis, only medication type showed a significant effect on the incidence of acute endophthalmitis (IRR, 1.80; 95% CI, 1.16–2.79; p = 0.01) (Table 3).
Discussion
In this study, we analyzed the incidence and risk factors of acute endophthalmitis after intravitreal anti-VEGF injections in patients with neovascular AMD by using the KNHIS health claims data and neovascular AMD registration codes from 2010 to 2019. Acute endophthalmitis after intravitreal anti-VEGF injection occurred very rarely (0.019%), and the risk of developing endophthalmitis was higher with aflibercept injections than with ranibizumab injections (IRR, 1.80; 95% CI, 1.16–2.79; p = 0.01).
Few studies have analyzed the rate of endophthalmitis after intravitreal anti-VEGF injections in patients with wet AMD. The incidence of endophthalmitis in our study differed in comparison with the findings reported in previous studies, which seems to be mainly related to differences in study design, patient characteristics, and the definition of endophthalmitis. In a 2-year single-center retrospective study, the incidence of clinically diagnosed endophthalmitis was reported to be 0.014%, which is consistent with our study [12]. However, another 4-year multicenter retrospective study found that the incidence of presumed infectious endophthalmitis was much higher (0.04%) [7]. Also, a single-center prospective study reported an incidence as high as 0.083% [6]. Similar to our study, these studies targeted patients who were clinically suspected of having infectious endophthalmitis after intravitreal anti-VEGF injection, received vitreous tap, and underwent intravitreal antibiotic injection or vitrectomy. However, unlike our study, these studies included patients with other retinal diseases, such as diabetic macular oedema, proliferative diabetic retinopathy, and retinal vein occlusion other than wet AMD, which may have affected the overall incidence of endophthalmitis. In particular, diabetic patients are vulnerable to infection due to innate immune suppression and blood circulatory dysfunction, which may affect the incidence of endophthalmitis after intravitreal injection [7,13].
The incidence of endophthalmitis according to the medications used also differed between studies. In this study, the incidence was slightly higher with intraocular injections of aflibercept (0.025% vs. 0.014%). In a study using US claims data, the incidence of antibiotic-associated endophthalmitis was higher with aflibercept than with ranibizumab (0.44% vs. 0.22%), and the risk of developing endophthalmitis was also higher with aflibercept (adjusted relative ratio, 2.04; 95% CI, 1.42–2.92; p < 0.05) [14]. However, some studies showed no statistically significant differences in the incidence of endophthalmitis by medication type [12,15,16].
The higher incidence of endophthalmitis after intravitreal aflibercept injection is difficult to explain. One possible explanation is that patients with severe sterile intraocular inflammation were also included in our analysis, which may have increased the incidence of acute endophthalmitis requiring invasive treatment. Aflibercept contains an Fc domain that can act as a proinflammatory factor and can cause sterile intraocular inflammation [17]. In patients with wet AMD, the Fc-gamma receptor IIa (FcγRIIa) is widely expressed throughout the retina [18]. Unlike ranibizumab, aflibercept, which has a higher molecular weight, does not permeate the retina through the intercellular space and is absorbed by neuronal cells and the retinal pigment epithelium [19]. Therefore, the Fc domain of aflibercept can bind to FcγRIIa, which is upregulated in wet AMD patients and ultimately induces inflammation [14,18].
Several potential factors are associated with endophthalmitis following intravitreal anti-VEGF injections. The most common cause may be bacterial or fungal contamination during the injection procedure. In a survey by Green-Simms et al. [20], 88% of US retinal specialists did not use a sterile drape, and only 33% wore sterile gloves. These practices may have increased the risk of endophthalmitis. However, antibiotic prophylaxis does not affect the incidence of endophthalmitis [21,22]. Environmental effects, such as septic injection techniques or contaminated aerosolized droplets, are another potential source of infection. An extremely low incidence of endophthalmitis (0.000%–0.008%) was reported when the procedure was performed in an operating room setting [23–26]. No difference was observed in the incidence rate of endophthalmitis following injections in the operating room and office (odds ratio, 3.06; 95% CI, 0.07–139.75), but the rate of development of culture-positive endophthalmitis was much higher following injection in the office (odds ratio, 21.52; 95% CI, 2.39–193.55) [27]. These results show that a sterile environment can contribute to reducing endophthalmitis after injection.
The incidence of endophthalmitis may also increase due to mishandling of the medication, i.e., problems with storage or preparation. A nationwide cohort study performed in France showed a higher risk of endophthalmitis with aflibercept than with prefilled ranibizumab (IRR, 1.80; 95% CI, 1.24–2.61). Antigenic particles derived from protein degradation or silicone oil droplets may form during rapid freezing, thawing, or storage outside the recommended temperature range of 2°C to 8°C [28]. In addition, during the process of transfer from a vial to a syringe, the medication may be exposed to the infection source. For patients treated with ranibizumab, prefilled syringes also showed a lower risk of endophthalmitis than nonprefilled syringes (IRR, 1.60; 95% CI, 1.14–2.25), which means that fewer manipulations and preparation under sterile environments are important to reduce postoperative endophthalmitis [4].
Our study had several limitations. First, the identification of cases with intravitreal anti-VEGF injections, endophthalmitis, and other comorbidities by using healthcare claims and KCD and KEDI codes might be inaccurate when compared with information obtained from medical charts. In particular, this method shows a greater potential for misclassification or underdetection-related bias. Second, the diagnosis of acute endophthalmitis was based on clinical suspicion rather than a strict bacteriological definition, and both culture-positive and culture-negative cases were included in the study. As a result, patients with severe sterile inflammation were also included in our analysis. Third, the lack of clinical information regarding factors such as medical history, visual acuity, and subtypes of neovascular AMD is another inherent limitation. Despite its low risk, endophthalmitis is a vision-threatening disease; therefore, an understanding of the visual prognosis of endophthalmitis after injection is important. Fourth, bevacizumab, which is less expensive than aflibercept or ranibizumab, is often intravitreally injected when the eligibility criteria for neovascular AMD are not met. However, since bevacizumab is an off-label drug, it is not covered by insurance and is excluded from the KNHIS database. Therefore, the incidence of acute endophthalmitis during intravitreal bevacizumab injections was not analyzed. Fifth, although the use of prefilled syringes may affect the incidence of endophthalmitis, it was not analyzed as a risk factor in this study. This is because the domestic approval date of the prefilled ranibizumab was June 2019, and prefilled aflibercept has not been marketed in Korea. In other words, prefilled syringe was used very limitedly within the study period, so it was not possible to compare the incidence of endophthalmitis between prefilled syringe and vials. In future research, analysis of syringe type should be considered as one of the risk factors for endophthalmitis.
Nevertheless, our study also had the following strengths. This is the largest retrospective claims data-based study of acute endophthalmitis after intravitreal anti-VEGF injection in patients with neovascular AMD in South Korea over a 10-year study period. Using this population-based database, we provided a reliable assessment of the incidence of acute endophthalmitis after intravitreal injection— an extremely rare complication—that was consistent with previous studies. We also demonstrated the safety of frequently performed procedures in neovascular AMD.
In this study, we demonstrated a generally low incidence of acute endophthalmitis after intravitreal anti-VEGF injections, consistent with previous studies. However, because neovascular AMD requires repeated intravitreal anti-VEGF injections, attention should be paid to the risk of endophthalmitis.
Acknowledgements
None.
Notes
Conflicts of Interest: None.
Funding: This work was supported by a National Health Insurance Ilsan Hospital grant (No. NHIS-2021-1-265). The authors alone are responsible for the content and writing of this manuscript.