Lower Energy to Make a Corneal Flap with a 60 kHz Femtosecond Laser Reduces Flap Inflammation and Corneal Stromal Cell Death But Weakens Flap Adhesion
Article information
Abstract
Purpose
To compare corneal flaps created in rabbits with a 60 kHz femtosecond (FS) laser using different levels of raster energy and to measure early inflammation, corneal stromal cell death, and late postoperative adhesion strength.
Methods
Sixty rabbits were divided into three groups of 20 each. A flap 110 µm thick and 9.0 mm in diameter was made in one eye of each rabbit at raster energies of 0.7 µJ, 1.1 µJ, and 2.4 µJ. Histopathological evaluation for inflammation and apoptosis using terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) staining was performed at 4 and 24 hours after flap creation. The adhesion strength of the flaps was measured with a tension meter at 1 and 3 months.
Results
Twenty four hours after flap creation, the 2.4 µJ group had more inflammatory and CD11b-positive cells than the 0.7 and 1.1 µJ groups. The number of TUNEL-positive cells increased with raster energy at 4 and 24 hours. The grams of force (gf) needed to detach the flaps at 3 months was significantly higher in 2.4 µJ group (170 gf) than in 0.7 µJ group (97.5 gf) and 1.1 µJ group (100 gf, p = 0.03).
Conclusions
Using raster energy lower than 1.1 µJ to make a flap with a 60 kHz FS laser decreases inflammatory cell infiltration and corneal stromal cell death in the central cornea but may result in a weaker flap than using higher raster energy (2.4 µJ).
Laser in situ keratomileusis (LASIK) has become the most frequently performed type of corneal refractive surgery for the correction of myopia [1]. Flap-related complications occur in about 5% of LASIK eyes with a microkeratome and can include off-center flaps, free flaps, irregular flap edges and stromal bed surfaces, epithelial abrasions, buttonholes, and flap lacerations [2-4]. Using a femtosecond (FS) laser for flap creation results in greater accuracy in intended flap thickness, better flap dimensions, and a lower frequency of flap-related complications than using a mechanical microkeratome [5-9]. LASIK patients have lower incidences of epithelial injury, faster recovery of corneal sensation, better astigmatic neutrality, a lower degree of spherical aberration, and better contrast sensitivity with the FS laser than with the mechanical microkeratome [10,11].
A 15 kHz FS laser was found to create a stronger flap than a mechanical microkeratome because it induced more inflammation in the flaps [7]. Introduction of the 60 kHz FS laser markedly reduced postoperative flap edema and diffuse lamellar keratitis compared with 15 kHz lasers [12]. Recently, de Medeiros et al. [13] reported that higher raster and side cut energy levels increased cell death and inflammatory cell infiltration when a 60 kHz FS laser was used to create corneal flaps. To our knowledge, the effect of raster energy level on the adhesion strength of corneal flaps created with a 60 kHz FS laser has not been determined.
We therefore confirmed the effect of raster energy levels on early inflammatory cell infiltration and corneal stromal cell death and compared the late adhesion strength of rabbit corneal flaps created with a 60 kHz FS laser setting at different raster energy levels.
Materials and Methods
Animals and preparation
Procedures were conducted in conformity with the ARVO Statement for the Use of Animals in Ophthalmic and Vision Research. Sixty male New Zealand white rabbits weighing 2.0 kg were divided into three equal groups. Each rabbit was anesthetized intramuscularly with tiletamine-zolazepam (10 mg/kg; Zoletil 50, Virbac Lab, Carros Cedex, France) and xylazine (Rompun 2%; Bayer Korea, Seoul, Korea) and had 0.5% proparacaine hydrochloride topical eye drops applied to each eye (Alcaine; Alcon-Couvreur, Puurs, Belgium) with 0.1% levofloxacin (Cravit; Santen Pharmaceutical Co., Osaka, Japan). Each right eye was proptosed anterior to the eyelids and temporarily retained in that conformation by clamping the temporal upper and lower eyelids together with a mosquito clamp.
Femtosecond laser procedure
We utilized a 60 kHz FS laser (60 kHz IntraLase FS laser; Abbott Medical Optics Inc., Santa Ana, CA, USA) to make corneal flaps 9.0 mm in diameter and 100 µm thick in the right eye of each rabbit. The head of each anesthetized rabbit was held still and a flat, glass contact lens was used to applanate the cornea without using a suction ring. Pulse spacing was 8-µm, and we used raster energies of 0.7 µJ (n = 20), 1.1 µJ (n = 20), and 2.4 µJ (n = 20) [12]. We compared flap inflammation and corneal stromal cell death in 10 eyes per group, as described previously [7]. Ofloxacin ointment (Tarivid, Santen Pharmaceutical Co.) was applied over the wound and tarsorrhaphy was performed in every eye for the first 4 or 24 hours. The rabbits were sacrificed at 4 and 24 hours after making a flap, thus, there were 5 eyes per group for each time point.
Measurement of flap inflammation: histology and immunohistochemistry
After H&E staining, the number of inflammatory cells in the corneal stroma around the flap was counted by a single observer in three fields of three tissue slides per specimen. The Kruskal-Wallis test was used to compare the differences among groups.
Expression of CD11b, a monocyte marker, was analyzed by immunohistochemistry (IHC) on paraffin-embedded cornea tissues; 4 µm thick sections from the paraffin blocks were used for IHC with an Envision-HRP detection system (Dako, Carpinteria, CA, USA). After deparaffinization and antigen retrieval by a pressure cooker in 0.01 M sodium citrate buffer (pH 6.0) at full power for 10 minutes, tissue sections were treated with 3% H2O2 for 5 minutes. The anti-CD11b antibody (1:50; Novus Biologicals, LLC, Littleton, CO, USA) was added to the slides and incubated at 4℃ overnight. The slides were treated with the Envision reagent for 30 minutes, and then sequentially incubated with diaminobenzidine chromogen for 10 minutes, counterstained with Meyer's hematoxylin, and mounted. Careful rinses with several changes of tris-buffered saline-0.1% tween buffer were performed at each step. A rat IgG1 isotype was used as a negative control to exclude the primary antibody. The slides were evaluated and photographed using a Leica Microphoto FXA microscope (Leica Microsystems Inc., Bannockburn, IL, USA).
Measurement of corneal stromal cell death: TUNEL assay
To evaluate the degree of apoptosis, terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) assays were performed with a fluorescein apoptosis detection system (Roche, Mannheim, Germany) according to the manufacturer's instructions. The tissues were deparaffinized by boiling in 0.1 M citrate buffer (pH 6.0) in a microwave for 15 minutes. Positive and negative control slides were included in each assay. Four sections from each specimen were evaluated under light microscopy at higher magnification (×400), with TUNEL-positive keratocytes around the flap counted by the same observer as above.
Measurement of adhesion strength
Adhesion strength was determined for 10 eyes per group. After waiting 5 minutes to allow the absorption of microcavitation bubbles, a spatula was pushed into the laser wound in the temporal quadrant near the hinge to separate the flap. The flap was then gently repositioned using the spatula and three 10-0 nylon sutures were used to reduce the detachment of the flap at the nasal, temporal and inferior interfaces. Only ofloxacin ointment was applied over the wound and tarsorrhaphy was performed in every eye for the first 24 hours. At 1 week after making a flap the attachment of flap was examined, and 10-0 nylon sutures were stitched out under the microscopy after the rabbits were anesthetized as previously mentioned.
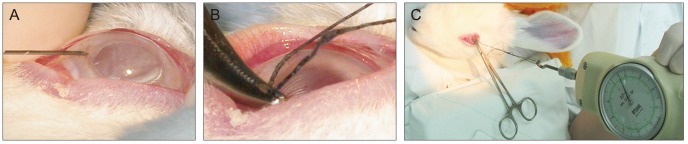
We attempted to find the edge of each flap and separate at least a portion of it to allow it to be gripped by a curved mosquito clamp for 6-0 black silk loop suturing under microscopy (Fig. 1A and 1B). A tension meter (Attonic, Aichi, Japan) was used to measure the adhesion strength at months 1 and 3, and flap movement was observed under microscopy (Fig. 1C).
Statistical analyses
Data are expressed as the mean ± SE. All statistical analyses were performed using SPSS ver. 12.0.1 (SPSS Inc., Chicago, IL, USA). At a particular time point, the numbers of inflammatory cells and apoptotic cells were compared between the three groups using ANOVA. Within each group, we compared the values at 4 and 24 hours using Student's t-test. Adhesion strengths were analyzed with non-parametric Mann-Whitney tests. Statistical significance was defined as p < 0.05.
Results
Histology, immunohistochemistry, and TUNEL assays
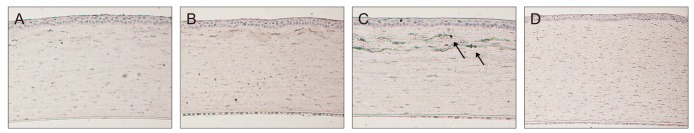
The number of inflammatory cells visible with H&E staining in the central cornea was significantly greater in all three groups at 4 and 24 hours than in the control eyes (p < 0.05) (Table 1). At 4 hours, inflammatory cell infiltration at the central cornea did not differ significantly among the three groups. At 24 hours, however, infiltration at the central cornea was significantly greater in the 2.4 µJ group than in 0.7 and 1.1 µJ groups (p < 0.05). Infiltration at the central cornea decreased significantly over time only in 1.1 µJ group (p = 0.30, 0.04, and 1.00 in groups 1, 2, and 3; respectively) (Table 1 and Fig. 2). IHC using anti-CD11b antibodies also showed that the 2.4 µJ group had more CD11b-positive cells than the 0.7 and 1.1 µJ groups at 24 hours (p < 0.05) and the number of CD11b-positive cells decreased significantly over time only in the 1.1 µJ group (p < 0.05). This is consistent with the results obtained by H&E staining (Table 1 and Fig. 3).
The number of infiammatory cells detected by H&E staining, CD11b-postitive cells, and TUNEL-postitive cells around the corneal flap

H&E staining of a central cornea 24 hours after flap creation in the 0.7 µJ (A), 1.1 µJ (B), 2.4 µJ (C), and control (D) groups. Inflammatory cell infiltration into the central cornea (arrows) was observed by light microscopy under a high power field (×400). At 24 hours, the 2.4 µJ group had more inflammatory cell in filtration than the other groups.
Immunohistochemistry of a central cornea using anti-CD11b antibodies 24 hours after flap creation in the 0.7 µJ (A), 1.1 µJ (B), 2.4 µJ (C), and control (D) groups. It also showed the 2.4 µJ group had more CD11b-positive cells than the 0.7 and 1.1 µJ groups at 24 hours (p < 0.05) and the number of CD11b-positive cells decreased significantly over time only in the 1.1 µJ group (p < 0.05), which were consistent with the findings on H&E staining (×400).
We also observed significantly more TUNEL-positive cells in the central cornea in each group at 4 and 24 hours than in the control eyes (p < 0.05) (Table 1 and Fig. 4). At 4 hours, TUNEL-positive cell infiltration was significantly greater in the 2.4 µJ group than in the 0.7 and 1.1 µJ groups (p < 0.05) and was significantly greater in the 1.1 µJ group than in the 0.7 µJ group (p < 0.05). At 24 hours, TUNEL-positive cell infiltration was significantly greater in the 2.4 µJ group than in the 0.7 µJ group. TUNEL-positive cell infiltration decreased significantly over time in the 1.1 and 2.4 µJ groups (p = 0.35, 0.05, and 0.05 in the 0.7, 1.1, and 2.4 µJ groups; respectively).

Terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) assay of a central cornea at 4 hours after flap creation in the 0.7 µJ (A), 1.1 µJ (B), and 2.4 µJ (C) groups and at 24 hours in the 0.7 µJ (E), 1.1 µJ (F), 2.4 µJ (G), and control (H) groups (×400). 4 and 24 hours after flap creation. TUNEL-positive cells (red arrow) were scattered throughout the areas anterior and posterior to the lamellar interface created by the femtosecond laser.
Adhesion strength
In the 0.7 µJ group, 65.6 ± 8.6 and 88.0 ± 12.8 grams of force (gf) were needed to separate the flaps at 1 and 3 months after flap creation, respectively; the corresponding values were 55.0 ± 15.0 and 100.0 ± 18.2 gf in the 1.1 µJ group and 73.0 ± 8.6 and 176.0 ± 15.0 gf in the 2.4 µJ group, respectively. The adhesion strength increased significantly over time only in the 2.4 µJ group (p = 0.18, 0.11, and 0.01 in 0.7, 1.1, and 2.4 µJ groups, respectively). The adhesion strength at 1 month was not higher in the 2.4 µJ group than in the 0.7 and 1.1 µJ groups (p = 0.52), but was significantly higher at 3 months (p = 0.03) (Fig. 5).

Measurements of adhesion strength using a tension meter. Adhesion in the 2.4 µJ group was significantly stronger than in the 0.7 and 1.1 µJ groups at 3 months (p = 0.04). The adhesion strength increased significantly over time only in the 2.4 µJ group. *Significant differences between groups at particular raster energy (p < 0.05). †Significant differences between at 4 and 24 hours (p < 0.05).
Discussion
The 60 kHz FS laser is one of the most widely used instruments for LASIK surgery. Recommended raster energies fall in the range of 0.8 to 1.0 µJ for spots and 8 × 8 to 9 × 9 µm for line separations [14]. When the raster energy was set lower (0.4 or 0.7 µJ), a 60 kHz FS laser could create LASIK flaps safely, although some areas in the flaps had scattered, incomplete tissue bridges [14]. Conversely, in order to reduce the incidence of flap tear during sub-Bowmann's keratomileusis with an FS laser it has been recommended that the raster energy be increased to slightly more than the minimum required (0.91 µJ) [15]. To date, however, little has been known about the effect of different raster energy levels on early inflammatory cell infiltration or the adhesion strength of corneal flaps.
In measuring inflammatory and apoptotic cell infiltration in the central cornea, we did not lift flaps after their creation with the 60 kHz FS laser as done in previous studies [12,13]. In a pilot study, however, we lifted the flaps after 4 and 24 hours and counted the numbers of inflammatory and apoptotic cells. The results were inconsistent among the three groups (data not shown), perhaps because incomplete tissue bridges between the flap and residual bed could be broken if we lifted the flap, which resulted in inconsistent inflammatory and apoptotic cell infiltration around the flaps. In addition, when the flaps are lifted, there might be a relatively uncontrolled entrance of inflammatory cytokines from the epithelium into the flap interface. Inflammatory cytokines can chemoattract inflammatory cells, resulting in apoptosis [16]. To remove the possible effect of incomplete tissue bridges and infiltrating cytokines, the flaps were not lifted after their creation. However, to measure adhesion strength we had to lift the flaps and gently reposition them using a spatula to simulate a realistic surgical setting.
Consistent with de Medeiros et al. [13], we found lower raster energy levels resulted in less inflammatory cells and less corneal stromal cell death. In terms of the TUNEL assay, we also had fewer TUNEL-positive cells in all three groups than de Medeiros et al.'s. This discrepancy could be due to the different raster energy levels used in each group, methods of tissue fixation, tissue sectioning, efficacy of the TUNEL assay, block type, and laser and experimental setting were different between the two studies.
Diffuse lamellar keratitis is attributed to endogenous factors resulting from epithelial injury during flap creation [17] or from necrotic debris due to direct energy-related effects of the FS laser [12,18]. It was noteworthy that inflammatory cell infiltration into the flaps made with a 60 kHz FS laser was quite rare at 4 and 24 hours after a flap was created with the highest raster energy (2.4 µJ). Following the introduction of the 60 kHz FS laser, the raster energy required to create a flap was dramatically reduced. Diffuse lamellar keratitis may no longer be a concern in 60 kHz FS laser-assisted LASIK, reducing the need to use strong topical steroids postoperatively. Nevertheless, it is noticeable that using lower raster energy to make a flap may result in a weak adhesion between the flap and residual bed. We may have to maintain the raster energy about 1.1 µJ to retain proper adhesion strength of flap. However, overestimation of our results should be avoided, because the ideal adhesion strength to secure the corneal flap after FS laser-assisted LASIK was not determined. In this study three groups were compared according to the raster energy level used to create a flap. Furthermore, the inverted side-cut design of flaps could be another option to strengthen adhesion [19].
Contrary to our results, a comparison of the adhesion strengths of corneal flaps created with a 150 kHz IntraLase FS laser set at 0.8 or 1.6 µJ side-cut energy showed no difference 75 days after flap creation [19]. Their study differed from ours in that they used a 150 kHz IntraLase FS laser instead of a 60 kHz FS laser and set the spot and line separation less than the 8 × 8 µm used in our study. Furthermore, in that study they divided the experimental groups according to side-cut energy instead of raster energy, and they compared both acute and inverted side-cut angles, suggesting that the inverted side cut could improve side cut wound apposition. Finally, the method the earlier group used to measure adhesion strength was different from ours, in that they glued the tension meter to the flap, whereas we found the edge of the flap and separated at least a portion of it, allowing it to be gripped by a curved mosquito clamp for 6-0 black silk loop suturing.
This study had several limitations. First, we were unable to determine the mechanism by which higher raster energy resulted in stronger flap adhesion, though we think that more inflammation induces stronger flap adhesion. Immunohistochemical staining and longer follow-ups are needed to clarify the relationship between strong flap adhesion and high raster energy. Second, laser ablation was not performed on the stroma and postoperative topical steroids were not applied after the creation of flaps, which were not clinically realistic omissions. Third, three sutures were necessary to reduce the detachment of the flap for a week seemed to increase the wound healing response and augment the flap adhesion strength compared to using no sutures. This does not affect the comparison in adhesion strength between flaps with sutures made by the different energy levels, but does affect the comparison between flaps with sutures and those without sutures.
In conclusion, a 60 kHz FS laser with a raster energy lower than 1.1 µJ resulted in less inflammatory cell infiltration and corneal stromal cell death in the central cornea, but resulted in weaker flaps than using higher raster energy (2.4 µJ). Even at higher raster energy, postoperative topical steroids may not be required due to only mild levels of inflammatory cell infiltration.
Acknowledgements
This study was supported by Basic Science Research Program through the National Research Foundation of Korea funded by the Ministry of Education, Science and Technology (NRF-2010-0025662) and a grant (2011-464) from the Asian Institute for Life Sciences in Seoul, Korea.
Notes
This article was presented as a scientific paper in 2009 ASCRS meeting at San Francisco, USA.
No potential conflict of interest relevant to this article was reported.