A Case of Pediatric Idiopathic Intracranial Hypertension Presenting with Divergence Insufficiency
Article information
Abstract
An 11-year-old female presenting diplopia only at distance was found to have comitant esotropia of 20 prism diopters (PD) at distance and normal alignment at nearer proximity. Other ocular movement, including abduction, was normal and a thorough neurologic examination was also normal. The deviation angle of esotropia was increased to 35 PD in 6 months, and a brain magnetic resonance imaging with venogram at that time demonstrated no intracranial lesion. A lumbar puncture showed increased opening pressure but the cerebrospinal fluid composition was normal. The patient was diagnosed as having idiopathic intracranial hypertension and treated with oral acetazolamide. Three months after treatment, the deviation angle decreased to 10 PD. This is a case report of divergence insufficiency in pediatric idiopathic intracranial hypertension, with an increasing deviation angle of esotropia. Although sixth cranial nerve palsy is a common neurologic manifestation in intracranial hypertension, clinicians should be aware of the possibility of divergence insufficiency. Also, ophthalmoparesis may not be apparent and typical at first presentation, as seen in this case, and therefore ophthalmologists should be aware of this fact, while conducting careful and proper evaluation, follow-up, and intervention.
Divergence insufficiency is a clinical sign characterized by esotropia at distance with normal abduction and no ocular misalignment at nearer proximity. Patients with divergence insufficiency thus complain of diplopia only at distance [1,2]. Diplopia due to divergence insufficiency has been described in several neurologic diseases such as encephalitis, neurobrucellosis, Miller Fisher syndrome, and also intracranial hypertension [3-6]. We encountered a case of divergence insufficiency in an 11-year-old girl with idiopathic intracranial hypertension (IIH), in which the angle of deviation increased with time. This publication reports the detailed findings of this case.
Case Report
An 11-year-old female patient visited the ophthalmology clinic with a four-month history of diplopia, especially when concentrating on a distant target such as letters on the whiteboard in school. She denied any headache during those periods. The patient had no remarkable past medical history. She had already visited another ophthalmology clinic with the same symptoms and underwent brain magnetic resonance imaging (MRI), which she was told was normal.
At the first visit, her visual acuity was 20 / 20 in both eyes. Slit lamp examination was done and there were no specific indications. Extraocular movement testing revealed 20 prism diopters (PD) comitant esotropia at distance, and straight at nearer proximity. Cycloplegic refraction was also done and the refractive error was -3.25 sph -0.50 cyl Ax180° in both eyes.
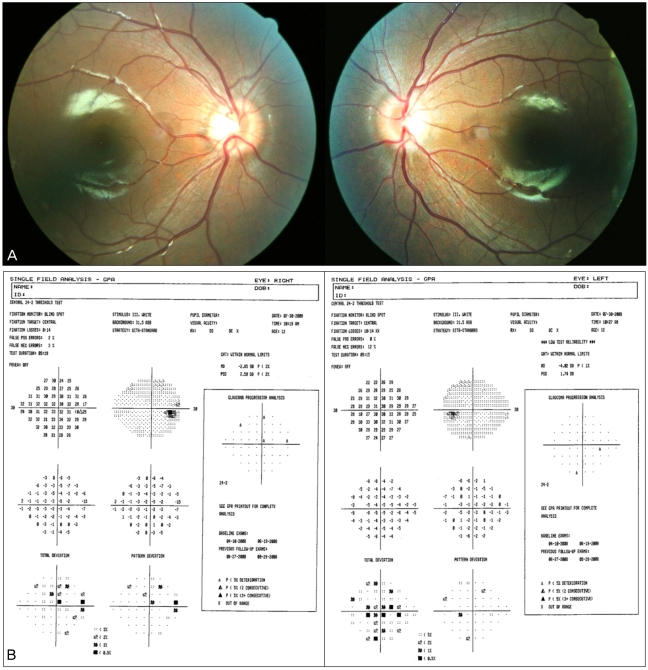
On fundus examination, mild optic disc edema was found in both eyes (Fig. 1A). Optical coherence tomography also demonstrated a mild thickening of the nerve fiber layer. A visual field test using the Swedish interactive thresholding algorithm standard 30-2 revealed a bilateral enlargement of the blind spot (Fig. 1B).

Ocular manifestation at presentation. There is mild bilateral optic disc swelling (A) and blind spot enlargement, according to a visual field test (B).
A systemic examination revealed no significant abnormality. At the time of initial presentation, the patient was obese with a body mass index of 29.7 kg/m2 (body weight 66 kg, height 149 cm). We asked the patient to bring the previous brain MRI for verify the findings, but she was lost to follow-up over the next four months. After four months, she returned to the clinic with an increased deviation angle of esotropia to 25 PD at the primary position, and still straight at nearer proximity. Optic disc edema, and bilateral enlargement of the blind spot in the visual field remained the same.
At six months after initial presentation, the deviation angle increased to 35 PD at distance without an abduction deficit. We suspected IIH, although there was no headache, normally typical and the most common symptom of pediatric IIH, and the patient underwent the brain MRI with venogram again. The brain imaging study ruled out any intracranial lesion including sinus thrombosis.
We referred the patient to pediatric neurology for a lumbar puncture. The cerebrospinal fluid (CSF) examination revealed a clear and colorless fluid, and an increased opening pressure of 230 mm H2O with a normal CSF composition. A diagnosis of IIH was made and the patient was started on a treatment of 750 mg acetazolamide a day. We also referred the patient to obesity clinic for body weight control.
Three months after treatment, her body mass index was reduced to 28.8 kg/m2 (body weight 64 kg, height 149 cm), and there was a progressive improvement of symptoms. The deviation angle of esotropia was reduced to 10 PD at distance and her eyes remained straight at nearer proximity (Fig. 2). Although the optic disc edema had not resolved (Fig. 3A), the blind spot enlargement had decreased slightly, according to a visual field test (Fig. 2B).

There was change in deviation angle after three months of treatment. The deviation angle of esotropia was decreased to 10 prism diopters at distance, and straight at nearer proximity. No abduction deficit was noted.
Discussion
IIH is a diagnosis of exclusion characterized by elevated intracranial pressure and normal CSF composition, in the absence of intracranial lesions or other underlying systemic causes [7]. The diagnostic characteristics of IIH were first enumerated by Dandy in 1937 [8] and were later formulated into a set of diagnostic criteria (modified Dandy criteria) by Smith in 1985 [9]. The typical patient is an obese but otherwise healthy woman of child-bearing age. According to Gordon [10], there has been an increasing incidence of IIH among adolescents (12 to 15 years old) as compared to young children (2 to 12 years old). Although pediatric IIH is now increasing in incidence, there is still no specific diagnostic criteria for pediatric IIH.
Pediatric IIH is further divided into prepubertal and pubertal IIH based on the development of secondary sexual characteristics of age. There are differences between those two groups. In prepubertal IIH, there is no sex predilection or tendency toward obesity. In pubertal IIH, female predominance has been found, with the percentage of affected males varying between 0% and 39%. Association with obesity is also found in pubertal IIH [11-14].
The course of pediatric IIH varies, and a child may present hours to several years after symptoms begin. Headache is the most common complaint with IIH and most children with IIH have headaches. However, there are also reports of IIH in the absence of headache, either because the child is too young to articulate symptoms or because headaches are completely absent. Nausea and vomiting are also known as classic symptoms of IIH. Patients may complain of blurry vision, diplopia, and stiff neck as well. Other reported symptoms of pediatric IIH include increasing head size, photophobia, anorexia, retro-orbital pain, lightheadedness, myalgia, head tilt, as well as a knee-chest position. Patients with IIH have a normal level of consciousness and functioning.
Sixth cranial nerve palsy was the only accepted neurological abnormality in diagnosing IIH until recently. Sixth cranial nerve palsy was documented in only 12% of adults, but it continues to be more common among children with IIH, occurring in 9% to 48% [14-17]. Other cranial nerve palsies, including the 3rd, 4th, 7th, 9th, and 12th cranial nerves, have been reported in children. Comitant esotropia worse at distance but without an obvious abduction defect may also be seen [16].
Divergence insufficiency was also reported in patients with intracranial hypertension who complain of diplopia. The divergence insufficiency pattern of esotropia was first described by Parinaud and Duane [18-21]. The unique indication of divergence insufficiency produces either confused vision or double vision at distance. Near vision is not disturbed. The diplopia always has a subtle onset, but may be intermittent or seldom emergent. In divergence insufficiency, the uncrossed diplopia should be measured the same in every field of gaze. There should be no limitation of abduction to the right or left [1,2,21].
Divergence insufficiency must be differentiated from other conditions that cause comitant esotropia at distance. Convergence spasm, decompensated esophoria, accommodative spasm, and bilateral sixth nerve palsy must be considered. Sudden development of confused distance vision with anxiety, headache, nausea, and dizziness are more likely to be divergence paralysis or sixth cranial nerve palsy. Sixth cranial nerve palsy exhibits incomitant strabismus. Esotropia is generally variable in spasm of near reflex. Patients with decompensated esophoria have esotropia of the same degree at distance and nearer proximity.
The exact mechanism of divergence insufficiency or sixth cranial nerve palsy has not been identified clearly, but several ideas have been suggested. One suggestion is that ophthalmoparesis in IIH may be related directly to very elevated CSF pressure and may be secondary to altered CSF flow in the posterior fossa [22]. Others have suggested that there might be involvement of the sixth cranial nerve, probably nuclear or infranuclear, in patients with divergence insufficiency. The sixth nerve nuclei lie in an area of the brain stem which is supplied by the terminal branches of the circumflex and paramedian perforating branches of the basilar artery. With increased intracranial pressure, critical perfusion levels may be reached such that divergence insufficiency suddenly appears, and that on reduction of intracranial pressure single vision is restored when perfusion of the nuclei exceeds this critical level [23]. In addition, Jamposky [24] expressed the view that divergence insufficiency does not exist and that all such patients actually have a bilateral sixth nerve palsy.
In our case, the patient had comitant esotropia without abduction limitation, and complained of binocular diplopia at distance without headaches. During the follow-up period, the deviation angle increased and after a brain MRI with venogram and lumbar puncture, she was diagnosed with IIH. Her ocular symptoms could be mistaken for the sixth cranial nerve palsy accompanying IIH. However, comitant esotropia was apparent only at distance and no ocular misalignment was found at nearer proximity, and other ocular movements, including abduction, was normal, which are all typical features of divergence insufficiency. Unusual feature of this patient was the increment of deviation angle with time. It might be presumably due to the sixth nerve paresis superimposed upon the pre-existing divergence insufficiency in its aggravating course of IIH.
In this report, we presented an unusual case of IIH presenting with divergence insufficiency, which featured an increasing deviation angle of esotropia with time. Although sixth cranial nerve palsy is the most common neurologic manifestation in IIH, clinicians should be aware of the fact that ophthalmoparesis in IIH can be variable and careful evaluation of each patient is required.
Notes
No potential conflict of interest relevant to this article was reported.