 |
 |
| Korean J Ophthalmol > Volume 24(1); 2010 > Article |
Abstract
Purpose
To assess the risk factors for endothelial cell loss after phacoemulsification with implantation of intraocular lens according to anterior chamber depth (ACD).
Methods
This prospective study included 94 eyes of 94 patients undergoing phacoemulsification cataract surgery. To assess the risk factors for corneal endothelial cell loss, we examined seven variables at 1 day, 1 week, 6 weeks, and 12 weeks postoperatively in each ACD-stratified group.
Results
Multiple linear regression analysis showed that the only variable influencing the percentage decrease in corneal endothelial cell density throughout the postoperative follow-up period in the long ACD group (ACD III) was nucleosclerosis. The variables influencing the percentage decrease in corneal endothelial cell density in the short ACD group (ACD I) at one day and one week postoperatively were corneal incisional tunnel length and nucleosclerosis.
Past reports have shown that several preoperative and intraoperative parameters may influence the risk of endothelial cell loss after phacoemulsification. Risk of endothelial cell damage increases with a high nucleus grade, advanced age, long phacoemulsification time (Phaco time), high ultrasound energy, small pupil diameter, large infusion volume, type of intraocular lens (IOL), and short axial length [1-7]. Additionally, phacoemulsification surgery is performed in a limited, confined space. Adequate space can lessen the damage induced by the phacoemulsification procedure. We also believe that risk factors could variably influence the corneal endothelium in different anterior chamber depth (ACD) conditions; even though the variable (ACD) does not affect the degree of endothelial cell damage, the ACD can influence the effect of other variables on the endothelium. In this study, we examined the effects of several preoperative and intraoperative parameters on corneal endothelial cell density after phacoemulsification in different ACD-stratified groups.
We also paid particular attention to a new variable, corneal incisional tunnel length, and its role in postoperative endothelial cell loss in ACD-stratified groups.
We prospectively examined 94 eyes of 94 patients scheduled to undergo phacoemulsification surgery.
Exclusion criteria included the following: a history of previous ocular surgery or inflammation, glaucoma, corneal pathology, trauma, and intraoperative complications such as posterior capsule rupture or postoperative complications.
All procedures were performed by the same surgeon using retrobulbar anesthesia. A 3.0 mm clear corneal incision was made in the superior quadrant. A capsulorhexis approximately 5.0 mm in diameter was created with forceps, then cortical cleaving hydrodissection was performed. The nucleus was emulsified using the stop and chop technique. After irrigation and aspiration of the cortex, a foldable acrylic IOL (SN60WF; Alcon Laboratories Inc., Fort Worth, TX, USA) was implanted in the bag. The same irrigating solution (balanced salt solution, BSS) and the same type of ophthalmic viscosurgical device (OVD, sodium hyaluronate 1.2%) were used for all patients.
Preoperatively, axial length (mm) and ACD (mm) were recorded using ultrasound A scanning (Compact II device; Quantel Medical Inc., Bozeman, MT, USA). Eyes were stratified into groups based on ACD as follows: ACD I, 1.5<ACDŌēż2.5 mm; ACD II, 2.5<ACDŌēż3.5 mm; ACD III, 3.5<ACDŌēż4.5 mm. Nuclear opacity was graded from 1 to 4 using the Lens Opacities Classification System. To evaluate central corneal endothelial cell density, specular microscopic photographs of the central corneal endothelium were taken using a non-contact specular microscope (noncon robo; Konan Medical Inc., Hyogo, Japan). We analyzed a minimum of 40 endothelial cells to calculate the endothelial cell density. Corneal endothelial cell loss was evaluated by measuring the percentage decrease in endothelial cell density of the central cornea (cells/mm2).
The percentage decrease in central corneal endothelial cell density was expressed as: (preoperative central corneal endothelial cell density-postoperative central corneal endothelial cell density)├Ś100/preoperative central corneal endothelial cell density.
Intraoperatively, we recorded phaco energy: (phacoemulsification time (seconds)├Śphacoemulsification power (%), total surgical time (min) and volume of irrigating solution (balanced salt solution, ml) used.
The corneal incisional tunnel length (mm) was measured with calipers (Fig. 1). Several factors affect the tunnel length, including the sharpness of the blade, the angle of approach of the blade and the thickness of the corneal tissue [8, 9]. The tunnel length was graded from 1 to 3 as follows: Grade 1, less than 1 mm; Grade 2, 1.0 to less than 2.0 mm; Grade 3, 2.0 to less than 3.0 mm.
Postoperatively, specular microscopic photographs of the central corneal endothelium were taken using a non-contact specular microscope at postoperative day 1, week 1, week 6, and week 12.
Statistical analyses were conducted using SPSS ver. 11.5 (SPSS Inc., Chicago, IL, USA). We compared percentage decreases in endothelial cell density using a one-way analysis of variance (ANOVA) according to ACD group. Univariate association of the seven variables (age, axial length, nucleosclerosis, tunnel length, operation time, irrigation volume, and phaco energy) with a decrease in endothelial cell density was evaluated by simple regression analysis. A multiple linear regression analysis with a stepwise regression was used to determine the best-fitting set of variables contributing to decreases in endothelial cell density.
p-values <0.05 were considered statistically significant.
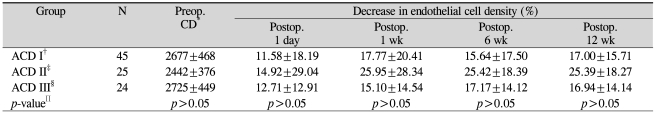
Before ACD stratification of all eyes, neither ACD nor axial length was associated with percentage decrease in endothelial cell density at 1 day, 1 week, 6 weeks, and 12 weeks postoperatively (p>0.05).
The percentage decrease in central endothelial cell density at 1 day, 1 week, 6 weeks, and 12 weeks postoperatively was not significantly different among the ACD-stratified groups (p>0.05) (Table 1).
Table 2 shows the distribution of seven variables in the ACD stratified group. The long ACD group (Group ACD III) showed a longer axial length than ACD Groups I and II.
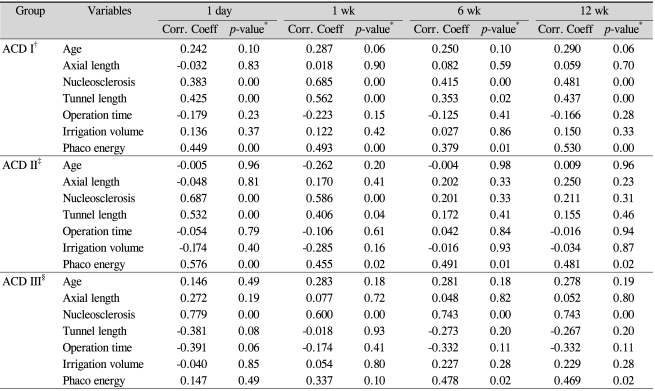
Table 3 shows the correlations of the seven variables on univariate analysis with the percentage decrease in central endothelial cell density at postoperative day 1, week 1, week 6, and week 12 in the ACD-stratified groups.
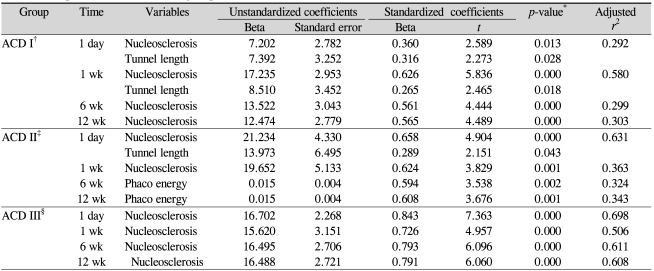
Table 4 shows a stepwise multiple regression analysis model to identify the best set of independent predictors of percentage decrease in corneal endothelial cell density at 1 day, 1 week, 6 weeks, and 12 weeks postoperatively in the ACD-stratified groups.
As shown in Table 3, on simple regression analysis, the significant factors for percentage decrease in central endothelial cell density at postoperative day 1 were nucleosclerosis (r=0.383, p=0.000), tunnel length (r=0.425, p=0.000) and phaco energy (r=0.449, p=0.000). The significant factors at postoperative week 1 were nucleosclerosis (r=0.685, p=0.000), tunnel length (r=0.562, p=0.000) and phaco energy (r=0.493, p=0.000). The significant factors at postoperative week 6 were nucleosclerosis (r=0.415, p=0.000), tunnel length (r=0.353, p=0.02) and phaco energy (r=0.379, p=0.01). The significant factors at postoperative week 12 were nucleosclerosis (r=0.481, p=0.00), tunnel length (r=0.437, p=0.00) and phaco energy (r=0.530, p=0.00).
Stepwise multiple linear regression analysis identified nucleosclerosis and tunnel length as independent predictors of a decrease in endothelial cell density at postoperative day 1, accounting for 29% of the variation in endothelial cell density decreases (Table 4). At week 1, nucleosclerosis and tunnel length were independent predictors of decreases of endothelial cell density, accounting for 58% of the variation in decrease of endothelial cell density. Nucleosclerosis alone was identified as an independent predictor of endothelial cell density decrease at postoperative week 6, accounting for 29% of the variation in decrease of endothelial cell density. Finally, at week 12 postoperatively, nucleosclerosis was still an independent predictor of endothelial cell density decrease, accounting for 30% of the variation in decrease of endothelial cell density.
As shown in Table 3, on simple regression analysis, the significant factors influencing decrease of corneal endothelial cell density at postoperative 1 day were nucleosclerosis (r=0.687, p=0.000), tunnel length (r=0.532, p=0.000) and phaco energy (r=0.576, p=0.000). The significant factors at postoperative week 1 were nucleosclerosis (r=0.586, p=0.000), tunnel length (r=0.406, p=0.04) and phaco energy (r=0.455, p=0.02). Phaco energy was the significant factor at postoperative week 6 (r=0.491, p=0.01). The significant factor at postoperative week 12 was also phaco energy (r=0.481, p=0.02).
Stepwise multiple linear regression analysis was also used to identify independent predictors of endothelial cell density decreases (Table 4). At postoperative day 1, nucleosclerosis and tunnel length were independent predictors of decrease of endothelial cell density and accounted for 63% of the variation in cell loss. Thirty-six percent of cell loss at postoperative week 1 was caused by nucleosclerosis. At postoperative week 6, phaco energy accounted for 32% of cell loss, while phaco energy explained 34% of cell loss at week 12 postoperatively.
Simple regression analysis was used to identify the significant factors influencing endothelial cell density (Table 3). The most significant factors were as follows: postoperative day 1, nucleosclerosis (r=0.779, p=0.000); week 1, nucleosclerosis (r=0.600, p=0.000); week 6, nucleosclerosis (r=0.743, p=0.000) and phaco energy (r=0.478, p=0.02); week 12, nucleosclerosis (r=0.743, p=0.000) and phaco energy (r=0.469, p=0.02).
In Table 4, stepwise multiple linear regression analysis identified nucleosclerosis as an independent predictor of endothelial loss at 1 day, 1 week, 6 weeks, and 12 weeks postoperatively, accounting for 69%, 50%, 61%, and 60% of variation in decrease of endothelial cell density, respectively.
The normal thickness and transparency of the cornea are maintained by the barrier function and the active fluid pump of the corneal endothelium. Endothelial alterations are considered important parameters of surgical trauma and are also essential for estimating the safety of surgical techniques. Endothelial cell density decreases at a greater rate after cataract surgery than it does in healthy, previously unoperated corneas. Corneal distortion, irrigation solution turbulence, instrument-related mechanical trauma, nuclear fragments, IOL contact, and free oxygen radicals can cause corneal damage during cataract surgery [1]. Several preoperative and intraoperative parameters (high nucleus grade, advanced age, long phaco time, high ultrasound energy, short axial length, and surgical skill) are associated with an increased risk of endothelial cell damage after phacoemulsification [1-6]. Researchers still debate whether short axial length and short anterior chambers are risk factors for endothelial cell damage [2, 4, 7]. Some reports have indicated that neither short axial length nor a shallow anterior chamber is associated with an increased risk of endothelial cells loss, which is similar to our result [2, 4]. This finding contrasts with reports by Walkow et al. [7] and Storr-Paulsen et al. [6], who found that shorter ACD eyes had a significantly increased risk for endothelial cell loss.
Shallow ACD leads to phacoemulsification being performed closer to the endothelium and may therefore be associated with an increased risk of endothelial cell loss.
Although anterior chamber depth is not always proportional to axial length [8], there were positive correlations between ACD and axial length in our study (r=0.379, p=0.00).
Corneal tunnel length can influence the process of cataract surgery. During lens phacoemulsification, if the tunnel created is longer than necessary, the phacotip tends to work uphill involuntarily. This disadvantage may create a certain amount of corneal distortion with compromised visibility in long ACD eyes (such as myopes). Because the deepening of the anterior chamber is caused by positive pressure of the infusion line in myopic patients and is not counterbalanced by an adequate vitreal thresh, it is often necessary to use the phaco tip and other instruments in a more vertical position than normal [9]. In hyperopic eyes, the corneal endothelium may potentially be closer to the phacoemulsification tip than it would be in a normalized eye due to the crowding of the anterior chamber [10], so long corneal tunnel length is associated with an increased risk for endothelial cell loss due to involuntary uphill work. In contrast, in the process of incision sealing, short corneal tunnel length tended to be associated with wound leakage [11].
Generally, past studies have reported normalization of morphometric indices, such as cell size-variation coefficient, percentage of hexagonality and corneal thickness, three months after uneventful cataract surgery [12, 13]. In our study, we did not observe cell stabilization until 12 weeks postoperatively, and the percentage decrease of endothelial cell density was higher than that noted in previous reports [6, 7]. We thought this difference could be due to the larger volume of irrigation solution and the longer operative time in our study, as well as differing surgical skills.
In our study, we observed different effects of various factors on postoperative endothelial cell loss according to ACD at each follow-up time point. Regarding corneal endothelial cell density, in short ACD eyes, nucleosclerosis, corneal tunnel length and phaco energy affected endothelial cell density throughout the 12 week postoperative follow-up period. However, in long ACD eyes, nucleosclerosis affected endothelial cell density throughout the 12 weeks while phaco energy affected endothelial cell density at 6 and 12 weeks postoperatively. We also identified a new variable: corneal incisional tunnel length. In short ACD eyes, corneal tunnel length was a major risk factor that can affect the decrease of corneal endothelial cell density throughout a 12 week postoperative follow-up. In eyes with middle-range ACD, corneal tunnel length was a risk factor for endothelial cell density only on postoperative day 1 and week 1. In long ACD eyes, corneal tunnel length was not associated with a decrease of endothelial cell density.
Therefore, we noted that the risk factors for endothelial cell loss after phacoemulsification could vary according to ACD stratified group. Corneal incisional tunnel length, an intraoperative factor that can be influenced by the sharpness of the blade, the angle of approach of the blade and the thickness of corneal tissue can be a risk factor for endothelial cell loss [14, 15]. Therefore, short corneal tunnels are indicated to reduce early postoperative endothelial cell loss in short ACD eyes.
REFERENCES
1. Mencucci R, Ponchietti C, Virgili G, et al. Corneal endothelial damage after cataract surgery: microincision versus standard technique. J Cataract Refract Surg 2006;32:1351-1354.


2. O'Brien PD, Fitzpatrick P, Kilmartin DJ, Beatty S. Risk factors for endothelial cell loss after phacoemulsification surgery by a junior resident. J Cataract Refract Surg 2004;30:839-843.


3. Hayashi K, Hayashi H, Nakao F, Hayashi F. Risk factors for corneal endothelial injury during phacoemulsification. J Cataract Refract Surg 1996;22:1079-1084.


4. Pereira ACA, Porfirio F, Freitas LL, Belfort R Jr. Ultrasound energy and endothelial cell loss with stop-and-chop and nuclear preslice phacoemulsification. J Cataract Refract Surg 2006;32:1661-1666.


5. Dosso AA, Cottet L, Burgener D, Di Nardo S. Outcomes of coaxial microincision cataract surgery versus conventional coaxial cataract surgery. J Cataract Refract Surg 2008;34:284-288.


6. Storr-Paulsen A, Norregaard JC, Ahmed S, et al. Endothelial cell damage after cataract surgery: divide- and conquer versus phaco-chop technique. J Cataract Refract Surg 2008;34:996-1000.


7. Walkow T, Anders N, Klebe S. Endothelial cell loss after phacoemulsification: relation to preoperative and intraoperative parameters. J Cataract Refract Surg 2000;26:727-732.


8. Steinert RF. Cataract surgery: technique, complication and management. 2004. 2nd ed. Philadelphia, PA: WB Saunders; p. 357-358.
9. Buratto L, Buratto L, Buratto C. In: Buratto L, Osher R, Masket S, Cataract surgery in complicated cases. 2000. Thorofare, NJ: SLACK; chap. 6.
10. Fine IH, Hoffmann RS. In: Buratto L, Osher R, Masket S, Cataract surgery in complicated cases. 2000. Thorofare, NJ: SLACK; chap. 7.
11. Ernest PH, Lavery KT, Kiessling LA. Relative strength of sclera corneal and clear corneal incisions constructed in cadaver eyes. J Cataract Refract Surg 1994;20:626-629.


12. Beltrame G, Salvetat ML, Driussi G, Chizzolini M. Effect of incision size and site on corneal endothelial changes in cataract surgery. J Cataract Refract Surg 2002;28:118-125.


13. Lundberg B, Jonsson M, Behndig A. Postoperative corneal swelling correlates strongly to corneal endothelial cell loss after phacoemulsification cataract surgery. Am J Ophthalmol 2005;139:1035-1041.


Table┬Ā1
Percentage decrease in endothelial cell density and percentage increase in corneal thickness in the anterior chamber depth (ACD) stratified group





 PDF Links
PDF Links PubReader
PubReader Full text via DOI
Full text via DOI Full text via PMC
Full text via PMC Download Citation
Download Citation Print
Print




