 |
 |
| Korean J Ophthalmol > Volume 37(6); 2023 > Article |
|
Abstract
Purpose
To evaluate the role of performing photocoagulation up to ora serrata during vitrectomy in preventing recurrent vitreous hemorrhage (VH) in patients undergoing pars plana vitrectomy (PPV) for proliferative diabetic retinopathy (PDR).
Methods
This retrospective, nonrandomized study included 60 eyes from 60 patients who had undergone PPV for VH due to PDR. These patients were divided into two groups: group 1, those who underwent photocoagulation up to ora serrata using the scleral indentation technique during surgery; and group 2, those who did not undergo scleral indentation when photocoagulation and underwent photocoagulation up to vortex veins. Their hospital records were analyzed to investigate the recurrence rate of VH, the time until recurrence of VH after surgery, logarithm of the minimal angle of resolution (logMAR) best-corrected visual acuity (BCVA) measured before surgery and at 1, 2, and 3 years after surgery, and the occurrence of complications such as neovascular glaucoma (NVG) during follow-up.
Results
Group 1 exhibited lower recurrence rate of VH (2 of 30 [6.7%] vs. 10 of 30 [33.3%], p = 0.01) and lower occurrence of postoperative NVG (2 of 30 [6.7%] vs. 8 of 30 [26.7%], p = 0.038) compared with group 2. There were no statistically significant differences in logMAR BCVA measured at 1, 2, and 3 years between the two groups (at 1 year: 0.54 ┬▒ 0.43 vs. 0.54 ┬▒ 0.44, p = 0.954; at 2 years: 0.48 ┬▒ 0.47 vs. 0.55 ┬▒ 0.64, p = 0.235; at 3 years: 0.51 ┬▒ 0.50 vs. 0.61 ┬▒ 0.77, p = 0.200). Logistic regression analysis showed that among several factors that could affect recurrence rate of VH, only range of photocoagulation performed was a statistically significant factor (odds ratio, 0.119; 95% confidence interval, 0.022-0.659; p = 0.015).
Pars plana vitrectomy (PPV) is now recognized as an important treatment for complications of diabetic retinopathy and for preventing blindness in patients with diabetic retinopathy [1,2]. However, recurrence of vitreous hemorrhage (VH) after PPV has been reported as a relatively common complication after surgery with an occurrence rate of 12.0% to 59.9% [3-5]. Although successful operation is performed, recurrent VH can significantly affect visual rehabilitation. It may require additional surgical procedure to improve vision [6,7]. Several factors are known to influence occurrence of recurrent VH following PPV, such as blood retinal barrier injury following retinal surgery, new vessel (NV) or fibrovascular proliferation (FVP) from the vitreous base, incomplete removal of blood clot in the peripheral vitreous, and residual fibrovascular tissue [8-10]. Many patients experience recurrent VH despite thorough removal of FVP and blood clots in the peripheral vitreous skirt, suggesting that newly formed postoperative NV in the vitreous base might be a significant source of recurrent VH [8,11-13].
Panretinal photocoagulation (PRP) is recognized as the most representative and important treatment for diabetic retinopathy to prevent harmful effect of NV [14]. However, PRP in an outpatient department or commonly performed during PPV cannot be applied to whole retina with limited access to peripheral retina, causing frequent occurrence of recurrent VH or NV. Yan et al. [15] have reported that photocoagulation in a more extensive area of retina might further reduce the amount of angiogenic factors and minimize the occurrence of NV, along with results of several cases of recurrent VH due to insufficient photocoagulation. Aylward et al. [16] and Reddy et al. [17] have reported that more extensive photocoagulation could induce regression of NV.
Considering the correlation between the area where photocoagulation is applied and the incidence of VH, it is questionable that performing photocoagulation up to ora serrata could reduce angiogenic factors and presence of NV and prevent recurrent VH. At first, we performed photocoagulation up to the vortex vein ampullae when diabetic vitrectomy. However, during subsequent follow-up, we experienced a few recurrence cases of VH. Therefore, we decided to perform photocoagulation over a wider range, up to ora serrata, and this retrospective study was conducted to assess the effectiveness of performing photocoagulation up to ora serrata by scleral indentation during PPV in patients with VH due to diabetic retinopathy in preventing recurrent VH.
This retrospective, nonrandomized study was approved by Chuncheon Sacred Heart Hospital Institutional Review Board (No. CHUNCHEON 2023-01-015). It complied with the tenets of the Declaration of Helsinki. The requirement for informed consent was waived due to the retrospective nature of the study.
We retrospectively analyzed patients from March 2010 to February 2020 who were diagnosed with naive proliferative diabetic retinopathy (PDR) and VH at the Department of Ophthalmology, Hallym University Chuncheon Sacred Heart Hospital (Chuncheon, Korea), and were observed for more than 3 years after PPV by a single retina specialist (MCS). We defined recurrent VH as VH that obscured retinal vessels (grade 2 or above in the Diabetic Retinopathy Vitrectomy Study [18]) by indirect ophthalmoscope at 3 weeks follow-up after surgery during follow-up. Cases of persistent VH after the surgery were not considered to recurrent VH and excluded. We divided patients into two groups: 30 eyes from 30 patients who underwent photocoagulation up to ora serrata using the scleral indentation technique during surgery (group 1) and 30 eyes from 30 patients who did not undergo scleral indentation when photocoagulation and underwent photocoagulation up to vortex veins (group 2). Fig. 1A and 1B show postoperative wide-field fundus photographs of groups 1 and 2. Patients with a history of previous vitreoretinal surgery and ophthalmic diseases that could affect visual acuity other than diabetic retinopathy, such as age-related macular degeneration, glaucoma, corneal opacity, or uveitis, were excluded from this study. In all patients, preoperative intravitreal bevacizumab injection was performed only once, on the day before surgery. All surgeries were performed with a vitrectomy machine (DORC, Associate 2500), a chandelier lighting system (Alcon Chandelier Lighting System, Alcon), and a noncontact wide-angle lens (Resight, Carl Zeiss Meditec AG). Under successful retrobulbar anesthesia, three valved 23-gauge trocar cannulas were inserted at the beginning of surgery. In the case of phakic eyes, cataract surgery was performed before PPV. Phacoemulsification was performed through clear corneal incision, followed by intraocular lens implantation. The incision was closed by 10-0 nylon. Core vitrectomy, the creation of a posterior vitreous detachment, peripheral vitrectomy, vitreous base shaving, and endolaser with or without scleral indentation were then performed (Fig. 2). Scleral depressor was held by the surgeonŌĆÖs second hand and used to indent the sclera. Balanced salt solution was used as a vitreous substitute. In the case of presence of retinal tear or FVP, perfluoropropane (C3F8) gas or silicone oil was used under the surgeonŌĆÖs decision. At the end of surgery, sclerotomy sites were sutured by 8-0 Vicryl. Any intravitreal bevacizumab injection was not performed until VH recurred postoperatively. Secondary vitrectomy was performed if recurrent VH persisted despite several intravitreal injections of bevacizumab. Hospital records were analyzed to investigate the recurrence rate of VH, the time until recurrence of VH after surgery, the best-corrected visual acuity (BCVA) measured before surgery and at 1, 2, and 3 years after surgery, and the occurrence of complications such as neovascular glaucoma (NVG) during follow-up. Visual acuity was measured using the Snellen visual acuity table and converted to logarithm of minimal angle resolution (logMAR). When visual acuity could not be measured numerically, finger count defined as logMAR 1.8, Hand motion defined as logMAR 2.3, and no light perception defined as logMAR 3.0 were used. All statistical analyses were performed using IBM SPSS ver. 26.0 (IBM Corp). A p-value of less than 0.05 was defined as statistically significant.
Clinical characteristics of the patients in both groups are shown in Table 1. There was no statistically significant difference in age distribution of patients between the two groups. The mean age was 56.03 ┬▒ 13.24 years in group 1 and 53.20 ┬▒ 7.80 years in group 2, showing no significant difference (p = 0.318). The logMAR BCVA before surgery was 1.58 ┬▒ 0.65 in group 1 and 1.35 ┬▒ 0.74 in group 2, showing no significant difference (p = 0.204). There were eight patients (26.7%) with pseudophakic lens in group 1 and seven (23.3%) in group 2, showing no significant difference (p = 0.766). There were six FVP cases (20.0%) in group 1 and five (16.7%) in group 2, showing no significant difference (p = 0.739). Four eyes (13.3%) in group 1 and three eyes (10.0%) in group 2 were injected with C3F8 gas, showing no significant difference (p = 0.688). Operation time was 70.67 minutes in group 1 and 58.0 minutes in group 2; however, the difference was not statistically significant (p = 0.054). There was no significant difference in sex, prevalence of hypertension, dyslipidemia, or chronic kidney disease, duration of diabetes mellitus, hemoglobin A1c, or average follow-up period either (Table 1).
Two patients (6.7%) in group 1 and 10 patients (33.3%) in group 2 had recurrent VH after surgery, showing a statistically significant difference (p = 0.01). Postoperative NVG occurred in two patients (6.7%) in group 1 and eight patients (26.7%) in group 2, showing a significant difference (p = 0.038). Postoperative logMAR BCVA levels measured at 1, 2, and 3 years after surgery are shown in Table 2, showing no statistically significant differences between the two groups.
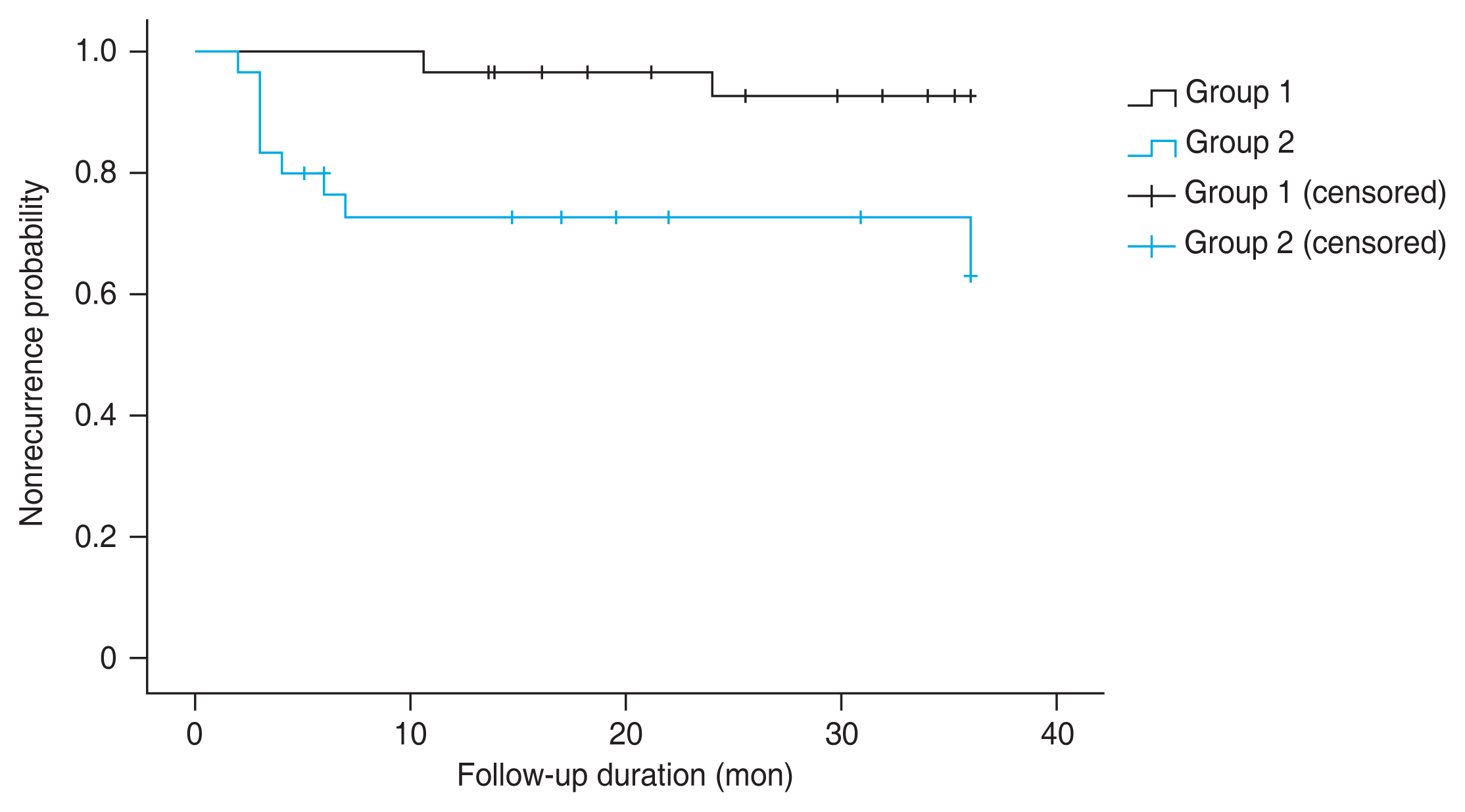
We performed logistic regression analysis to determine whether the recurrence of VH was significantly associated with other factors. The analysis showed that administering photocoagulation to ora serrata by scleral indentation was associated with a reduced recurrence rate of VH (odds ratio, 0.119; 95% CI, 0.022-0.659; p = 0.015). However, recurred VH showed no statistically significant associations with other factors such as presence of FVP, prevalence of hypertension, chronic kidney disease, or dyslipidemia, hemoglobin A1c level, operation time, and usage of gas tamponade (Table 3). Kaplan-Meier analysis was performed for recurrent VH (Fig. 3), showing a significantly lower recurrence rate of VH in group 1 than in group 2 (p = 0.038). Additionally, secondary vitrectomy was performed for one patient in group 1 and four patients in group 2. Ahmed valve implantation was performed for all NVG cases because of poor control of intraocular pressure.
Through this study, we found that the recurrence rate of VH decreased when photocoagulation up to ora serrata was performed during PPV for patients with VH due to PDR. PRP is accepted as an absolute treatment in diabetic retinopathy [14,19]. The possibility of maintaining a stable state without severe complications in the retina depends on how large area is photocoagulated completely [20,21]. However, several studies have reported cases in which NV progresses or does not sufficiently regress despite PRP [22,23]. These results might be because these studies were based on outpatient departments. PRP in outpatient department alone cannot be implemented sufficiently to prevent angiogenic effect fully, thus increasing the risk of complications such as recurrent VH and NVG.
Marra et al. [24] have conducted a retrospective study comparing combination of vitrectomy and endoscopic PRP up to ora serrata of retina and standard therapy including outpatient PRP and intravitreal bevacizumab in NVG patients. They reported that 91.7% of 27 eyes showed regression of NV of iris in patients who underwent endoscopic PRP up to ora serrata of retina, whereas 48.0% of 27 eyes showed regression of NV in the control group. Similarly, our results revealed a lower late of postoperative NVG in patients who underwent photocoagulation up to ora serrata than in patients who underwent photocoagulation up to vortex veins (2 of 30 [6.7%] vs. 8 of 30 [26.7%], p = 0.038), suggesting that photocoagulation over a wider range could prevent retina from falling into an ischemic state. However, their study differed from ours in that they did not target VH patients or investigate recurrence rates of VH according to the size of the area where photocoagulation was applied.
In our study, clinical characteristics between the two groups showed no statistically significant differences. The overall occurrence rate of postoperative VH was 12 of 60 (20.0%), including 2 of 30 (6.7%) in patients who underwent photocoagulation up to ora serrata (group 1) and 10 of 30 (33.3%) in patients who were sufficiently applied photocoagulation up to vortex veins (group 2), showing a statistically significant difference (p = 0.01) between the two groups. Of overall patients, the earliest recurrence period was 2 months. There was no case of early recurrent VH. This might be because remnant blood clot and FVP, which could be the main cause of early VH, were removed as much as possible even from the vitreous base. In addition, sclerotomy site suture was applied to all patients in our study. This method might prevent vitreous incarceration and lower occurrence of fibrovascular ingrowth (FVIG) at the sclerotomy site [25,26]. FVIG is known to be an important factor for the development of recurrent VH [8,15]. Therefore, the occurrence of FVIG at the sclerotomy site was sufficiently lowered in our patients.
Recurrent VH after PPV to treat diabetic retinopathy is one of the common complications. Although several studies have been conducted about recurrent VH, the recurrence rate of VH in postoperative period varies from study to study [3-5]. Several factors can affect the recurrence VH, including systemic risk factors (such as age, hyperlipidemia, and diabetic nephropathy [3,27,28]) and intraoperative factors [8-10].
Yeh et al. [29] have evaluated the effect of cryotherapy of peripheral retina and sclerotomy sites in preventing postoperative recurrent VH. They hypothesized that cryotherapy applied to sclerotomy sites might prevent progression of FVIG, causing a decreased rate of recurrent VH after PPV. In their study, the recurrence rate of VH was 3 of 26 (11.5%) in the group that underwent photocoagulation and anterior peripheral retinal cryotherapy and 1 of 23 (4.3%) in the group that underwent additional cryotherapy at sclerotomy site, similar to that in group 1 of the present study. However, cryotherapy might result in worse structural outcomes including retinal dragging and permanent scar change of retina, choroid or sclera and lower long-term visual acuity than photocoagulation [30]. Furthermore, as mentioned above, we sufficiently reduced occurrence of FVIG in sclerotomy site in our patients by applying sclerotomy site suture. Moreover, by performing 23-gauge vitrectomy in our study, we further lowered the possibility of FVIG in sclerotomy site compared to study by Yeh et al. [29], in which patients underwent a 20-gauge vitrectomy. According to these results and differences, our method is likely to be effective enough to lower the rate of recurrent VH using relatively simple procedures with fewer complications compared to cryotherapy.
Several studies have reported that certain factors are associated with postoperative recurrent VH. Yang et al. [31] have reported that intraocular gas tamponade with 10% C3F8 gas might be useful for proliferative diabetic retinopathy to reduce early recurrent VH. Baget-Bernaldiz et al. [32] have reported that preoperative presence of FVP can increase the chance of recurrent VH. We also compiled statistics about other factors that might affect the recurrence rate, including the usage of gas tamponade during surgery and presence of FVP. Results showed that the presence of FVP was associated with recurrence rate of VH with a marginal significance (p = 0.096). However, usage of gas tamponade did not show a statistically significant association (p = 0.812) with recurrent VH. This difference in results could be largely due to a relatively small number of patients, causing low statistical power and non-significant result. A further comprehensive study involving more patients is needed.
Other intraoperative manipulations for reducing postoperative VH have also been introduced. Landers and Perraki [33] have implicated that laser capsulotomy is likely to treat postoperative VH in patients who have previously undergone PRP and have intraocular lens with intact posterior capsule. Cheema et al. [34] have reported that removal of residual vitreous cortex is beneficial for reducing postoperative VH following diabetic vitrectomy. Qiu et al. [35] have concluded that intravitreal viscoelastic agent at the end of surgery can reduce postoperative VH after vitrectomy for PDR patients in their prospective study. We believe that, in addition to the above manipulations, performing photocoagulation up to ora serrata is an important technique in diabetic vitrectomy because it can inhibit angiogenic factors as much as possible for preventing recurrent VH.
Limitations of this study include the lack of randomization because of retrospective study, small number of subjects that could induce statistical error, and the possibility that selection bias might intervene in the choice of surgical method. It is necessary to conduct a comprehensive study with a greater number of patients in the future. In addition, our patients did not undergo postoperative wide-field fluorescein angiography, which was important to verify whether peripheral NV had regressed. Kim et al. [36] have actually investigated about this retrospectively and reported that peripheral NV observed in the postoperative widefield fluorescein angiography is significantly associated with the recurrence of VH. However, since we thoroughly minimized other causes of recurrent VH including FVIG in sclerotomy site and remnant blood clot or FVP, we believe that our results provide a reasonable basis for suppressed angiogenic factors, which are main causes of NV, even without confirmation of NV regression through postoperative fluorescein angiography.
In conclusion, our study is meaningful in that studies about the effectiveness of the method that performs photocoagulation up to ora serrata during PPV in patients with diabetic retinopathy have not been reported yet. Thus, photocoagulation treatment over a wider range with scleral indentation might be a useful way to preserve visual acuity without complications and further improve the quality of life of patients with PDR and VH.
References
1. Norton EW, Machemer R. A new approach to the treatment of selected retinal detachments secondary to vitreous loss at cataract surgery. Trans Am Ophthalmol Soc 1971;69:63-70.


2. Yang CM. Surgical treatment for diabetic retinopathy: 5-year experience. J Formos Med Assoc 1998;97:477-84.

3. Novak MA, Rice TA, Michels RG, et al. Vitreous hemorrhage after vitrectomy for diabetic retinopathy. Ophthalmology 1984;91:1485-9.


4. Schachat AP, Oyakawa RT, Michels RG, et al. Complications of vitreous surgery for diabetic retinopathy. II. Postoperative complications. Ophthalmology 1983;90:522-30.


5. Tolentino FI, Cajita VN, Gancayco T, et al. Vitreous hemorrhage after closed vitrectomy for proliferative diabetic retinopathy. Ophthalmology 1989;96:1495-500.


6. Brown GC, Tasman WS, Benson WE, et al. Reoperation following diabetic vitrectomy. Arch Ophthalmol 1992;110:506-10.


7. Han DP, Murphy ML, Mieler WF, et al. Outpatient fluid-air exchange for severe postvitrectomy diabetic vitreous hemorrhage: long-term results and complications. Retina 1991;11:309-14.


8. Hershberger VS, Augsburger JJ, Hutchins RK, et al. Fibrovascular ingrowth at sclerotomy sites in vitrectomized diabetic eyes with recurrent vitreous hemorrhage: ultrasound biomicroscopy findings. Ophthalmology 2004;111:1215-21.


9. West JF, Gregor ZJ. Fibrovascular ingrowth and recurrent haemorrhage following diabetic vitrectomy. Br J Ophthalmol 2000;84:822-5.



10. Lewis H, Abrams GW, Williams GA. Anterior hyaloidal fibrovascular proliferation after diabetic vitrectomy. Am J Ophthalmol 1987;104:607-13.


11. Koch FH, Kreiger AE, Spitznas M, et al. Pars plana incisions of four patients: histopathology and electron microscopy. Br J Ophthalmol 1995;79:486-93.



13. Hotta K, Hirakata A, Ohi Y, et al. Ultrasound biomicroscopy for examination of the sclerotomy site in eyes with proliferative diabetic retinopathy after vitrectomy. Retina 2000;20:52-8.


14. The Diabetic Retinopathy Study Research Group. Photocoagulation treatment of proliferative diabetic retinopathy: clinical application of Diabetic Retinopathy Study (DRS) findings, DRS report number 8. Ophthalmology 1981;88:583-600.


15. Yan H, Cui J, Lu Y, et al. Reasons for and management of postvitrectomy vitreous hemorrhage in proliferative diabetic retinopathy. Curr Eye Res 2010;35:308-13.


16. Aylward GW, Pearson RV, Jagger JD, et al. Extensive argon laser photocoagulation in the treatment of proliferative diabetic retinopathy. Br J Ophthalmol 1989;73:197-201.



17. Reddy VM, Zamora RL, Olk RJ. Quantitation of retinal ablation in proliferative diabetic retinopathy. Am J Ophthalmol 1995;119:760-6.


18. The DRVS Research Group. Two-year course of visual acuity in severe proliferative diabetic retinopathy with conventional management: Diabetic Retinopathy Vitrectomy Study (DRVS) report #1. Ophthalmology 1985;92:492-502.


19. Early Treatment Diabetic Retinopathy Study Research Group. Fundus photographic risk factors for progression of diabetic retinopathy: ETDRS report number 12. Ophthalmology 1991;98(5 Suppl):823-33.


20. Wand M, Madigan JC, Gaudio AR, et al. Neovascular glaucoma following pars plana vitrectomy for complications of diabetic retinopathy. Ophthalmic Surg 1990;21:113-8.


21. Jung CI, Kim SY, Kim SD. Comparison of surgical result of vitrectomy vs combined phacoemulsification and vitrectomy in patients with diabetic vitreous hemorrhage. J Korean Ophthalmol Soc 2000;41:2375-80.
23. Kim MK, Chung H. Effective dose of laser photocoagulation in proliferative diabetic retnopathy. J Korean Ophthalmol Soc 1998;39:111-8.
24. Marra KV, Wagley S, Omar A, et al. Case-matched comparison of vitrectomy, peripheral retinal endolaser, and endocyclophotocoagulation versus standard care in neovascular glaucoma. Retina 2015;35:1072-83.


25. Sabti K, Kapusta M, Mansour M, et al. Ultrasound biomicroscopy of sclerotomy sites: the effect of vitreous shaving around sclerotomy sites during pars plana vitrectomy. Retina 2001;21:464-8.


26. Kim IG, Lee SJ, Park JM. Comparison of the 20-gauge conventional vitrectomy technique with the 23-gauge releasable suture vitrectomy technique. Korean J Ophthalmol 2013;27:12-8.



27. Yoon JT, Kim CK, Sohn JH, et al. Systemic risk factors for postoperative vitreous hemorrhage following diabetic vitrectomy. J Korean Ophthalmol Soc 2001;42:434-40.
28. Thompson JT, Auer CL, de Bustros S, et al. Prognostic indicators of success and failure in vitrectomy for diabetic retinopathy. Ophthalmology 1986;93:290-5.


29. Yeh PT, Yang CM, Yang CH, et al. Cryotherapy of the anterior retina and sclerotomy sites in diabetic vitrectomy to prevent recurrent vitreous hemorrhage: an ultrasound biomicroscopy study. Ophthalmology 2005;112:2095-102.


30. Ng EY, Connolly BP, McNamara JA, et al. A comparison of laser photocoagulation with cryotherapy for threshold retinopathy of prematurity at 10 years: part 1. Visual function and structural outcome. Ophthalmology 2002;109:928-35.


31. Yang CM, Yeh PT, Yang CH. Intravitreal long-acting gas in the prevention of early postoperative vitreous hemorrhage in diabetic vitrectomy. Ophthalmology 2007;114:710-5.


32. Baget-Bernaldiz M, Romero-Aroca P, Mira-Puerto A, et al. Risk factors for recurrent vitreous hemorrhage in type 2 diabetes mellitus patients after posterior vitrectomy. J Clin Med 2023;12:2989.



33. Landers MB, Perraki AD. Management of post-vitrectomy persistent vitreous hemorrhage in pseudophakic eyes. Am J Ophthalmol 2003;136:989-93.


34. Cheema RA, Mushtaq J, Cheema MA. Role of residual vitreous cortex removal in prevention of postoperative vitreous hemorrhage in diabetic vitrectomy. Int Ophthalmol 2010;30:137-42.



Fig.┬Ā1
Postoperative wide-field fundus photography. (A) Fundus photography of group 1 patients showing that even the peripheral retina is photocoagulated. (B) Fundus photography of group 2 patients revealing photocoagulation up to vortex veins.

Fig.┬Ā3
Kaplan-Meier curve showing the period until recurrence of vitreous hemorrhage after surgery in group 1 (patients who underwent photocoagulation up to ora serrata) and group 2 (patients who underwent photocoagulation up to vortex veins).
Table┬Ā1
Clinical characteristics of patients (n = 60)
Table┬Ā2
Surgical outcome of patients (n = 60)
Table┬Ā3
Logistic regression analysis of other factors associated with recurrent vitreous hemorrhage (n = 60)
- TOOLS




 PDF Links
PDF Links PubReader
PubReader ePub Link
ePub Link Full text via DOI
Full text via DOI Full text via PMC
Full text via PMC Download Citation
Download Citation Print
Print




