 |
 |
| Korean J Ophthalmol > Volume 33(2); 2019 > Article |
Dear Editor,
Macular hole (MH) formation after anti-vascular endothelial growth factor therapy (VEGF) is a rare complication. Some cases of MH development after intravitreal bevacizumab have been reported, but there has been only one reported case of MH after intravitreal anti-VEGF for treatment of diabetic macular edema (DME). We report a patient who developed an MH after intravitreal bevacizumab injection for DME and MH closure after vitrectomy.
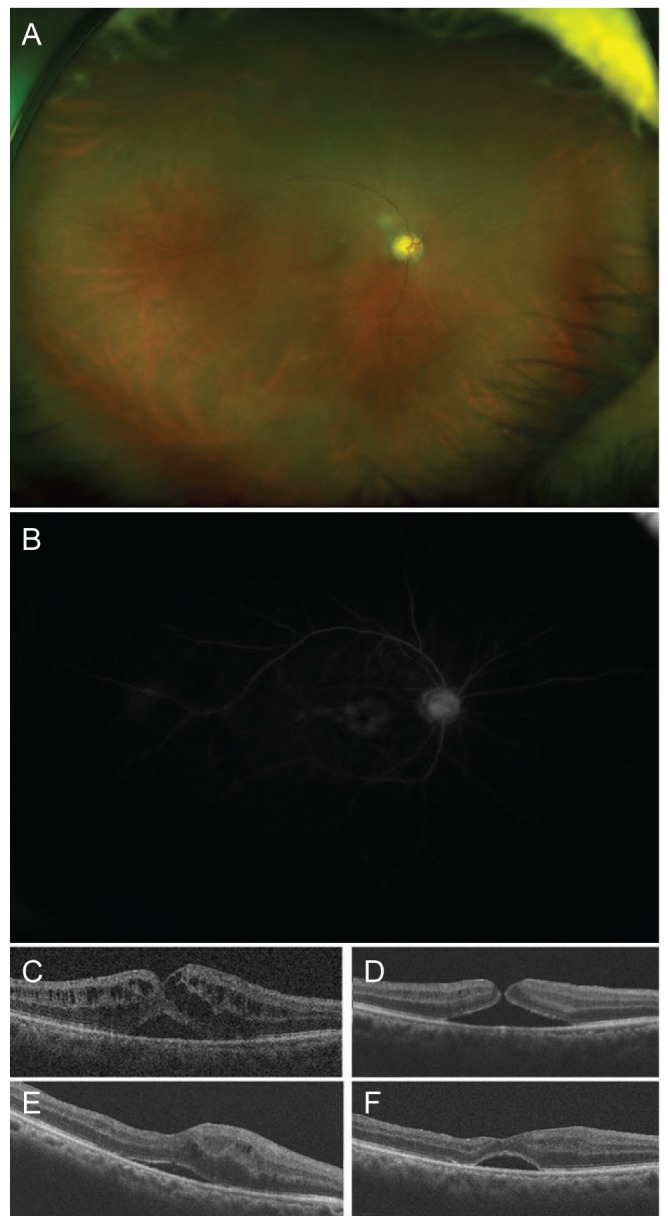
A 56-year-old male presented with non-proliferative diabetic retinopathy in the right eye. The best-corrected visual acuity (BCVA) was 20 / 200 in the right eye and 20 / 30 in the left eye. Intraocular pressure was 14 mmHg in the right eye and 15 mmHg in the left eye. Anterior segment findings were normal in the right eye. Fundus examination showed a few retinal hemorrhages in his right eye (Fig. 1A). Fluorescein angiography showed a diffuse petaloid pattern of leakage around the central fovea (Fig. 1B). Optical coherence tomography (OCT) revealed a thin epiretinal membrane, serous macular detachment and intraretinal edema that was located at the outer retina (Fig. 1C). After giving informed consent, the patient received an intravitreal 1.25 mg bevacizumab injection with a 30-gauge needle. Two weeks after injection, the BCVA in his right eye improved to 20 / 100. OCT revealed the formation of a full thickness MH and decreasing intraretinal edema (Fig. 1D). Four weeks after injection, the MH was still open. The patient underwent vitrectomy combined with cataract surgery and intraocular lens implantation. Internal limiting membrane peeling and gas tamponade with 20% sulfur hexafluoride were successfully performed. One month after surgery, OCT confirmed successful closure of the MH, but the serous macular detachment still remained (Fig. 1E). The BCVA in his right eye was 20 / 100. The patient refused further treatment due to economic reasons. Six months after vitrectomy, the serous macular detachment remained unchanged (Fig. 1F).
The responsible factors for MH formation after intravitreal anti-VEGF have been assumed to exist at the retinal pigment epithelium, retinal surface, and in the vitreous. Several potential mechanisms have been implicated to explain this process. Induction of vitreous incarceration following anti-VEGF injections could enhance vitreoretinal traction and subsequently MH development [1]. Chemical compounds introduced into the vitreous cavity and structural modification of the vitreous body following anti-VEGF therapy could also trigger incomplete posterior vitreous detachment (PVD), vitreomacular traction, and subsequent MH formation [2]. Grigoropoulos et al. [3] hypothesized that intravitreal injections can increase vitreomacular traction due to globe deformation during needle insertion and vitreous incarceration at the insertion site following treatment. This was proposed to cause vitreous syneresis and increase vitreofoveal traction leading to incomplete PVD, resulting in focal sites of traction on the retinal surface and MH formation. In our case, the PVD itself might not have been a causative factor for MH formation, because the PVD was induced with active aspiration during surgery. Liquefaction necrosis of the Müller cells and adjacent neural cells due to persistent ischemia leads to cystoid macular edema, a known cause of MH formation [4]. We postulated that intravitreal bevacizumab injection might have had an indirect role in the development of MH formation by favoring the rupture of distended Müller cells and intraretinal cysts. In this case, the coalescence and breakdown of large intraretinal cysts after bevacizumab injection in the presence of serous macular detachment could have caused MH. In addition, contraction of the thin epiretinal membrane and increased vitreomacular traction caused by the intravitreal injection could have contributed to the formation of MH.
In conclusion, we report a case of MH formation after intravitreal bevacizumab injection for treatment of DME. Although the occurrence of MH after intravitreal bevacizumab injection is uncommon, physicians should be well acquainted with this complication.
Acknowledgements
This study was supported by 2017 Research Grant from Kangwon National University (520170439).
Notes
Conflict of Interest: No potential conflict of interest relevant to this article was reported.
REFERENCES
1. Querques G, Souied EH, Soubrane G. Macular hole following intravitreal ranibizumab injection for choroidal neovascular membrane caused by age-related macular degeneration. Acta Ophthalmol 2009;87:235-237.

2. Raiji VR, Eliott D, Sadda SR. Macular hole overlying pigment epithelial detachment after intravitreal injection with ranibizumab. Retin Cases Brief Rep 2013;7:91-94.


Fig. 1
Ocular findings at initial presentation. (A) Fundus examination showed a few retinal hemorrhages. (B) Fluorescein angiography revealed a diffuse petaloid pattern of leakage around the central fovea. Serial changes in optical coherence tomography (OCT) image. (C) Before treatment, OCT revealed a thin epiretinal membrane, serous macular detachment, and intraretinal edema that was located at the outer retina. (D) Two weeks after the injection of bevacizumab, OCT showed the formation of a full thickness macular hole and decreasing intraretinal edema. (E) One month after vitrectomy, the macular hole was closed, but the serous macular detachment remained. (F) Six months after vitrectomy, the serous macular detachment remained unchanged



 PDF Links
PDF Links PubReader
PubReader Full text via DOI
Full text via DOI Full text via PMC
Full text via PMC Download Citation
Download Citation Print
Print




