Choroidal Thickness Indicates Subclinical Ocular and Systemic Inflammation in Eyes with Behçet Disease without Active Inflammation
Article information
Abstract
Purpose
To investigate whether subfoveal choroidal thickness, measured using enhanced depth imaging optical coherence tomography (EDI-OCT), is an indicator of subclinical ocular or systemic inflammation in eyes with Behçet disease (BD) without active ocular inflammation.
Methods
A retrospective analysis was used to examine clinical features of non-uveitic patients with BD (NUBD group), patients with a previous history of Behçet uveitis in an inactive state (IUBD group), and healthy controls were evaluated from October 2014 to September 2015. Subfoveal choroidal thickness was measured using EDI-OCT.
Results
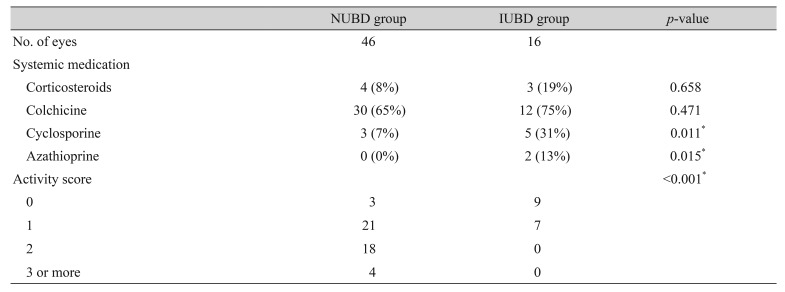
The NUBD group included 46 eyes in 24 patients; the IUBD group included 16 eyes in 11 patients; and the control group included 35 eyes in 23 individuals. The mean subfoveal choroidal thicknesses differed significantly among these groups. Choroidal thickness was significantly greater in the NUBD (310.5 ± 81.0 µm) than in the IUBD (263.1 ± 56.6 µm, p = 0.013) and control (256.9 ± 67.9 µm, p = 0.002) groups. The disease activity score was significantly higher in the NUBD than in the IUBD group (p < 0.001), while the use of cyclosporine was significantly associated with choroidal thickness in eyes with NUBD (p = 0.039).
Conclusions
Subfoveal choroidal thickness, as measured by EDI-OCT, may be a clinical indicator of subclinical ocular inflammation and systemic inflammation in BD patients without active ocular inflammation.
Behçet disease (BD) is a systemic immune-mediated vasculitis, affecting both arteries and veins in multiple organs [12]. Ocular involvement has been observed in 30% to 70% of BD patients, manifesting as anterior uveitis, posterior uveitis, panuveitis, and/or retinal vasculitis [134]. The severity of ocular BD seems to decrease over time, with less severe manifestations and better visual prognosis [356]. However, repeated involvement of the posterior segment may result in severe visual impairment.
Although the devastating ocular symptoms of BD in the fundus are caused by obliterative and necrotizing vasculitis, histopathological studies of the choroid have demonstrated diffuse and focal infiltration by inflammatory cells, including CD4+ T cells and macrophages [78]. In addition, imaging modalities, such as indocyanine green angiography, in patients with active Behçet uveitis (BU) have shown irregular filling of the choriocapillaris, choroidal filling defects, and dye leakage from choroidal vessels, suggesting involvement of the choroid in BD [910].
The development of enhanced depth imaging optical coherence tomography (EDI-OCT) has improved visualization of choroidal tissue, allowing choroidal involvement to be investigated noninvasively in many diseases. EDI-OCT has shown significantly increased choroidal thickness in BD patients with active uveitis [1112], as well as in the uninvolved eyes of patients with unilateral BU, compared with control subjects [11]. Because BD is a systemic vascular disease, these findings suggest that patients with BD and without any evidence of ocular involvement may still have subclinical choroidal involvement [11]. That is, the highly vascularized choroid is likely to be affected, regardless of whether clinical uveitis is present. Therefore, investigating choroidal changes in BD not only has potential significance for understanding the pathophysiology, but can also provide information about subclinical ocular inflammation, which may reflect systemic inflammation in BD.
This study was designed to investigate alterations in choroidal thickness in patients with BD without acute choroidal inflammation and to assess any relationship between systemic factors, such as systemic inflammation and immunosuppressant agents, with choroidal thickness. These findings may help determine whether choroidal thickness is a clinical indicator of subclinical ocular inflammation and systemic inflammation in BD patients without active ocular inflammation.
Materials and Methods
This study adhered to the tenets of the Declaration of Helsinki and was approved by the institutional review board of the Ajou University Hospital, Suwon, Korea (AJIRB-MED-MDB-15-452). The medical records of patients diagnosed with BD in the ophthalmology and/or dermatology departments of the Ajou University Hospital were retrospectively reviewed. Informed consent was waived due to the retrospective nature of the study. These patients met the diagnostic criteria for BD of both the International Study Group and the Research Committee of Japan [131415]. All patients underwent a comprehensive ocular examination and OCT imaging at baseline; patients with BU were also examined when the disease was inactive. Eyes with other retinal pathologies or any history of ocular surgery, as well as eyes with refractive errors greater than ±6.0 D, were excluded.
The BD patients without active ocular inflammation were divided into two groups based on their history of BU. The non-uveitic BD (NUBD) group consisted of patients with BD, but without any evidence or history of ocular manifestations. The inactive uveitic BD (IUBD) group consisted of BD patients with any history of uveitis, but in an inactive state for longer than three months. An inactive state was defined according to the Standardization of Uveitis Nomenclature working group classification as zero cells in the anterior chamber, with no signs of inflammation involving the posterior segment, including no signs of development of retinitis, vasculitis, papillitis, macular edema, or retinal hemorrhage [16].
Patients that underwent OCT imaging also underwent EDI-OCT simultaneously, according to our ophthalmology clinic protocol. Images were acquired using the Heidelberg Spectralis OCT (Heidelberg Engineering, Heidelberg, Germany) and were analyzed with Spectralis OCT software version 6.0. Two high-quality horizontal and two vertical line scans passing through the fovea were obtained within a 5 × 30-degree area at the fovea, with 100 scans averaged for each section. Two independent retinal specialists (YC and SJ) measured these parameters, and their mean data were used for statistical analysis.
Evaluation of systemic inflammation in patients with BD was limited to the items included in the Behcet's Syndrome Activity Scale, not the detailed scoring scale, due to the retrospective nature of this study [17]. The presence or absence of oral ulcers, genital ulcers, skin lesions, eye symptoms, gastrointestinal symptoms, vascular symptoms, and joint pain at the time of OCT examination and recorded in patients' medical records was noted, with each scored as 1 (presence) or 0 (absence) [17].
Numerical values were reported as the mean ± standard deviation. Baseline demographic characteristics and choroidal thicknesses in the NUBD, IUBD and control groups were compared using the chi-square test, Mann-Whitney U-test or Kruskal-Wallis test, as appropriate. A Pearson or Spearman correlation analysis was performed to examine the association between choroidal thickness and possible risk factors. The inter-observer reliability of the data measured by the two retinal specialists was expressed as the intraclass correlation coefficient. A p-value <0.05 was considered statistically significant. All statistical analyses were performed using PASW Statistics ver. 18.0 (SPSS, Chicago, IL, USA).
Results
The study evaluated 46 eyes in 24 patients with NUBD and 16 eyes in 11 patients with IUBD. The control group consisted of healthy subjects in our database, matched to patients in the other groups by age, spherical equivalents, and gender; this group consisted of 35 eyes in 23 subjects. There were no significant differences among the three groups in mean age, gender ratio, spherical equivalents and incidence of hypertension (Table 1).
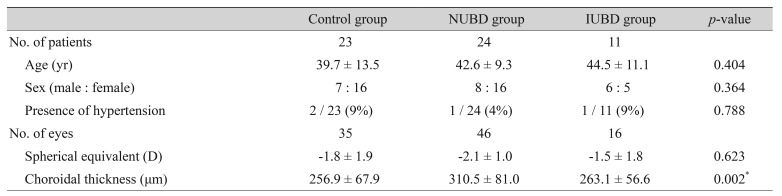
Demographic characteristics and subfoveal choroidal thickness in patients with Behçet disease, with or without uveitis, and control subjects
Of the 16 eyes in the IUBD group, 3 had previous anterior uveitis while 13 had either posterior uveitis or panuveitis. Mean choroidal thickness differed significantly among the three groups of eyes (p = 0.002), the value was significantly thicker in the NUBD (310.5 ± 81.0 µm) than in the IUBD (263.1 ± 56.6 µm, p = 0.013) and control (256.9 ± 67.9 µm, p = 0.002) groups (Fig. 1). The inter-observer reliability of the two retinal specialists who performed the measurements was excellent with an intraclass correlation coefficient of 0.955 for choroidal thickness (p < 0.001).

Subfoveal choroidal thicknesses in the three groups of eyes. Mean choroidal thickness was significantly greater in the non-uveitic Behçet disease (NUBD) than in the inactive uveitic Behçet disease (IUBD) and control groups.
Seven signs of systemic inflammation were evaluated, with each scored as 1 if present and 0 if absent; the systemic activity score was defined as the sum of these items and ranged from 0 to 7. The distribution of systemic activity scores is summarized in Table 2. Systemic activity scores differed significantly in the NUBD and IUBD groups (p < 0.001).
Higher percentages of patients in the IUBD than in the NUBD group were found to be taking cyclosporine (p = 0.011) and azathioprine (p = 0.015). In contrast, the use of corticosteroids and colchicine did not differ in these two groups (Table 2). Spearman correlation analysis showed that the use of cyclosporine was significantly associated with choroidal thickness (r = −0.263, p = 0.039), whereas none of the other medications were significantly correlated with choroidal thickness.
Discussion
BD is a systemic vascular inflammatory disease and the choroid is a highly vascularized tissue [218]. Choroidal involvement in BU has been well-documented both histologically and by conventional imaging modalities, such as indocyanine green angiography and fluorescein angiography [78910]. Retinochoroidal thickening has been detected in eyes with active BU by A-scan echography, suggesting a primary etiological role of choroidal inflammation in BU [19]. Based on these findings, several studies have used EDI-OCT to measure choroidal thickness in patients with BU. These studies found that increased choroidal thickness was associated with active ocular inflammation in patients with BD, and that thickness was reduced with the amelioration of ocular inflammation [1112]. In addition, subfoveal choroidal thicknesses of uninvolved pair eyes in patients with BD were significantly thicker than in control eyes [11]. Our study showed similar results. Mean choroidal thickness was significantly greater in the NUBD group than in the IUBD and control groups. Because BD is a systemic disease, patients with BD, but without any evidence of ocular involvement, may experience subclinical choroidal involvement due to systemic inflammation. In the same context, increased choroidal thickness in an acute phase of BU can be normalized by aggressively suppressing systemic inflammation. The percentages of patients that used cyclosporine and azathioprine were significantly higher, while the severity of systemic inflammation was lower, in the IUBD than in the NUBD group, suggesting that the systemic immunosuppression therapy used in the IUBD group also reduced choroidal inflammation and choroidal thickness. These findings indicate that choroidal thickness may be a clinical indicator of subclinical ocular inflammation, as well as a reflection of the severity of systemic inflammation. However, additional prospective studies that use detailed questionnaire surveys are needed to confirm the relationship between anti-inflammatory agents and choroidal thickness.
To analyze whether factors other than systemic inflammation were associated with choroidal thickness, we also investigated whether treatment with systemic immunosuppressants differed in the NUBD and IUBD groups. We found that treatment with cyclosporine, one of the immunosuppressants most widely used to treat BU [20], was associated with reduced choroidal thickness. Hypertension is a common side effect of cyclosporine use, which suggests that this agent affects vascular structure [2122]. The incidence of choroidal thickness is lower in patients with hypertension compared to those without, as high intravascular pressure leads to arteriolar sclerosis and vascular contraction in the choroid [23]. Furthermore, recent in vivo and in vitro experiments showed that cyclosporine had modulatory effects on endothelial and smooth muscle cells, leading to the contraction of smooth muscle cells and vascular structure collapse [2122]. We previously suggested that non-vascular smooth muscle cells (NVSMCs), which are abundant in the choroid, are responsible for changes in choroidal thickness in patients with central serous chorioretinopathy [24]. Choroid thickening in acute central serous chorioretinopathy is likely due to an increase in sympathetic input, reducing the number of NVSMCs and resulting in thinning of the choroid. Altogether, these findings suggest that use of cyclosporine may cause thinning of the choroid, directly by reducing the number of NVSMCs or indirectly by increasing intravascular pressure.
Our results indicate differences in choroidal thicknesses in patients with IUBD and control subjects which differ from previous findings [1125]. In our study, mean choroidal thickness did not differ significantly between the control eyes and IUBD eyes with a mean inactive period of 11.6 months. In contrast, a previous study found that choroidal thickness was significantly greater in quiescent BU eyes, with a mean inactive period of 7.0 months, compared with control eyes [11]. This difference may reflect the tendency of choroidal thickness to decrease over time during inactive periods. Alternatively, this discrepancy may be due to the different designs of the two studies, including differences in exclusion criteria and the definition of the inactive phase of BD. The previous study excluded eyes with spherical equivalent refractive errors greater than ±3.0 diopters, whereas this study excluded eyes with refractive errors greater than ±6.0 diopters. Furthermore, the lack of information about the use of systemic immunosuppressant agents in the previous study makes comparison of the two studies difficult, since specific immunosuppressant agents can affect choroidal thickness.
There were several major limitations to this study, including the retrospective design and the relatively small number of patients, all from a single center. The systemic activity score had to be evaluated retrospectively by reviewing medical records, therefore, this result could be underestimated due to insufficient recording. Furthermore, all subjects were of Korean ethnicity, suggesting that these findings may be inapplicable to patients of other ethnic or racial backgrounds.
This retrospective study demonstrated that subfoveal choroidal thickness was thicker in eyes with BD than in eyes with BU and control eyes. Systemic inflammation and treatment with aggressive immunosuppressant agents may contribute to alterations in choroidal thickness in patients with BD. These findings suggest that choroidal thickness may be a clinical indicator of subclinical ocular inflammation, which may also reflect the severity of systemic inflammation. Additional studies that evaluate a larger number of eyes and use systemic inf lammation scores to compare the same group of BD patients at different times during their course of disease may provide additional confirmation for our conclusions.
Notes
This study was presented as a poster at the 17th International Conference on Behçet's Disease on September 15–17, 2016 in Matera, Italy.
Conflict of Interest: No potential conflict of interest relevant to this article was reported.