 |
 |
| Korean J Ophthalmol > Volume 32(3); 2018 > Article |
Abstract
Purpose
To evaluate outcomes and factors associated with surgical failure in patients who underwent combined phacoemulsification and Ahmed glaucoma valve (AGV) implantation.
Methods
This retrospective and longitudinal study enrolled 40 eyes (38 patients) that underwent combined phacoemulsification and AGV implantation. Visual acuity, intraocular pressure (IOP), and number of antiglaucoma medications were evaluated preoperatively and postoperatively. Complete success was defined as a last follow-up IOP of 6 to 21 mmHg without medication, qualified success as an IOP of 6 to 21 mmHg with medication, and failure as an IOP of >21 or <6 mmHg.
Results
The mean follow-up period was 18 Âą 10 months. Preoperative diagnoses were chronic angle closure glaucoma (35.0%), neovascular glaucoma (22.5%), uveitic glaucoma (17.5%), primary open-angle glaucoma (15.0%), and other (10.0%). IOP decreased from a mean of 30.5 Âą 8.7 to 14.5 Âą 3.7 mmHg at the last follow-up visit (p < 0.001). Treatment was classified as qualified success in 18 eyes (45%), complete success in 15 (37.5%), and failure in seven (17.5%). Twenty-two eyes (55%) showed improvement in visual acuity. The most common postoperative complication was a transient hypertensive phase (five eyes, 12.5%). Tube-iris touch was associated with surgical failure (hazard ratio, 8.615; p = 0.008).
Cataract is the leading cause of blindness, and glaucoma is the leading cause of irreversible blindness worldwide [1,2]. Elderly patients with these two coexisting conditions are often encountered clinically [1,3]. Primary angle closure glaucoma caused by forward movement of the lens or phacomorphic or phacolytic glaucoma mandates the removal of the crystalline lens for the reduction of intraocular pressure (IOP). Phacoemulsification alone has a certain role in controlling IOP in primary angle closure glaucoma [4,5,6,7]; however, it is insufficient for control of IOP when angle closure persists and peripheral anterior synechiae forms [8,9,10]. Additional procedures, such as goniosynechialysis, EX-PRESS (Alcon Laboratories, Fort Worth, TX, USA) implantation, or trabeculectomy can be performed at the same time as phacoemulsification [7,11,12]. In some situations, phacotrabeculectomy may be inappropriate. In eyes with a history of failed filtering surgery; refractory glaucoma such as neovascular glaucoma (NVG), uveitic glaucoma, or secondary glaucoma; or previous bleb-related complications, success rates may be low [3,13]. Common complications after phacotrabeculectomy include hypotony, hyphema, and uveitis, among others [14,15]. Postoperative hypotony has been reported to be an independent risk factor for surgical failure of phacotrabeculectomy [14]. Furthermore, bleb-related infections can cause devastating visual loss [16].
The outcomes of combined phacoemulsification and glaucoma drainage implant (GDI) surgery were reported in several studies, with a relatively high success rate (range, 87.5% to 100%) [3,13,17,18]. However, the risk factors for failure of combined phacoemulsification and GDI surgery have not been investigated in depth. In this study, we therefore evaluated the surgical outcomes of combined phacoemulsification and GDI surgery and assessed the risk factors for poor prognosis.
The medical records of patients who underwent combined phacoemulsification and GDI surgery at Asan Medical Center between August 2011 and May 2016 were consecutively reviewed. Patients with a postoperative follow-up period of less than 6 months were excluded. A single glaucoma specialist (KRS) performed all surgeries using a repeatable and consistent operative technique in all the patients. The study was approved by the institutional review board of Asan Medical Center (2016-1282), Seoul, Korea. Informed consent was waived by the institutional review board due to retrospective design. The study followed the tenets of the Declaration of Helsinki.
An Ahmed-FP7 (New World Medical, Rancho Cucamonga, CA, USA) glaucoma valve (AGV) was used in all the patients. All of the intraocular lenses (IOLs) used were single-piece acrylic hydrophobic or plate-shaped hydrophilic lenses (each used in 50% of cases). In all cases, a single-stage procedure was performed and intraoperative antiproliferative agents such as 5-fluorouracil or mitomycin C were not used.
Initially, subconjunctival lidocaine injection was given either supratemporally or supranasally. A corneal traction suture was placed, and a fornix-based conjunctival pocket was created either supratemporally or supranasally. Although the supratemporal site was preferred, sites were chosen at the surgeon's discretion, depending on factors such as conjunctival scarring, presence of a previous tube, presence of peripheral anterior synechiae, and depth of the anterior chamber, which affect entry of the tube into the anterior chamber. To prevent excessive postoperative fibrosis, vessels were not cauterized during conjunctival dissection and scleral flap formation. After exposure of the scleral bed, which measures approximately 5 Ă 7 mm, a limbal-based partial-thickness autologous scleral flap was prepared using a Beaver blade. AGV was primed with balanced saline solution to confirm patency. 5-0 polypropylene (Prolene; Ethicon, Edinburgh, UK) thread was incorporated into the tube lumen. The tube was then ligated near the tube-plate junction with an 8-0 polyglactin suture (Vicryl, Ethicon), and the 5-0 Prolene thread was removed. Although AGV has a flow-restricting system consisting of two thin silicone elastomer membranes, early postoperative hypotony is frequently encountered after AGV implantation at rates as high as 19.4% [19]. Valve failure or aqueous leaking around the tube opening site in the early postoperative phase typically results in hypotony [20,21]. The effectiveness of partial ligation of the AGV tube for prevention of postoperative hypotony has been validated [20], so we routinely ligate the tube with absorbable thread to prevent early postoperative hypotony. The AGV plate was placed on the sclera approximately 8 to 10 mm behind the limbus and was secured to the sclera with 9-0 nylon. The tube was buried in the subconjunctival space either temporally or nasally in order to prevent obstruction of the surgical field during phacoemulsification. Standard clear corneal phacoemulsification and IOL implantation were performed. Some viscoelastic material was left in place to maintain the anterior chamber while the tube was inserted. The corneal incision was closed with a 10-0 nylon suture. The tube was then trimmed to an appropriate length and inserted into the anterior chamber through a 23-gauge needle tract beneath the scleral flap. The tube was fixed on the sclera with a 9-0 nylon suture. The scleral flap was closed with two 9-0 nylon sutures. Finally, the conjunctiva was reapproximated to the limbus with 8-0 Vicryl. No subconjunctival injections of antibiotics or dexamethasone were administered. Corticosteroid ointment and a pressure patch were applied at the end of the surgery. Topical antibiotics were prescribed 2 to 4 times per day for 4 weeks; topical steroids were prescribed 4 to 8 times per day for 4 weeks and tapered at the surgeon's discretion.
Preoperative data included age, sex, diagnosis, spherical equivalent (SE), axial length (AL), visual acuity (VA), IOP, and number of antiglaucoma medications. The primary outcome was IOP measured at postoperative months 1, 3, 6, 9, 12, and 24 using Goldmann applanation tonometry. Final IOP was defined as that from the most recent examination. Complete success was defined as IOP of 6 to 21 mmHg without medication, qualified success as IOP of 6 to 21 mmHg with medication, and failure as sustained IOP of >21 or <6 mmHg with or without medication on two or more visits [3,13,17,18]. Treatment was also considered a failure in patients who lost light perception after surgery or underwent further glaucoma surgery [17,18]. Postoperative VA, SE, number of antiglaucoma medications, and complications were also recorded. Absolute refractive error in diopters (D) was calculated using various IOL power calculation formulas.
Data are presented as mean Âą standard deviation or number and percentage. Preoperative and postoperative mean IOP and mean number of antiglaucoma medications were compared using paired Student's t-tests. The Friedman test was used to compare the predictability of various IOL power calculation formulas [22,23,24]. The Cox proportional hazards model was used to evaluate factors associated with failure. Hazard ratios (HRs) and 95% confidence intervals were calculated. Kaplan-Meier survival analysis was also performed. Cumulative probabilities of success were compared by the log rank test. All statistical analyses were performed with the PASW Statistics ver. 18.0 (SPSS, Chicago, IL, USA) statistical package. A p-value of <0.05 (two-tailed) was considered to indicate statistical significance.
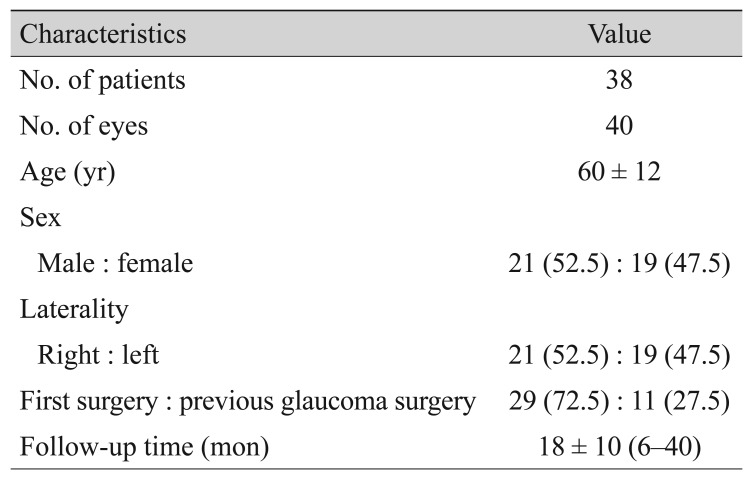
In total, 40 eyes of 38 patients were included in final analysis. All of the patients were Korean. Mean age was 60 Âą 12 years. The follow-up period ranged from 6 to 40 months, with a mean of 18 Âą 10 months. Demographic characteristics are summarized in Table 1. Twenty-nine eyes (72.5%) were primary cases and 11 eyes (27.5%) had undergone a previous glaucoma surgery. Preoperative diagnoses are listed in Table 2. Chronic angle closure glaucoma (CACG) was most common, which accounted for 14 eyes (35.0%), followed by NVG (nine eyes, 22.5%), uveitic glaucoma (seven eyes, 17.5%), primary open-angle glaucoma (POAG; six eyes, 15.0%), and other (four eyes, 10.0%).
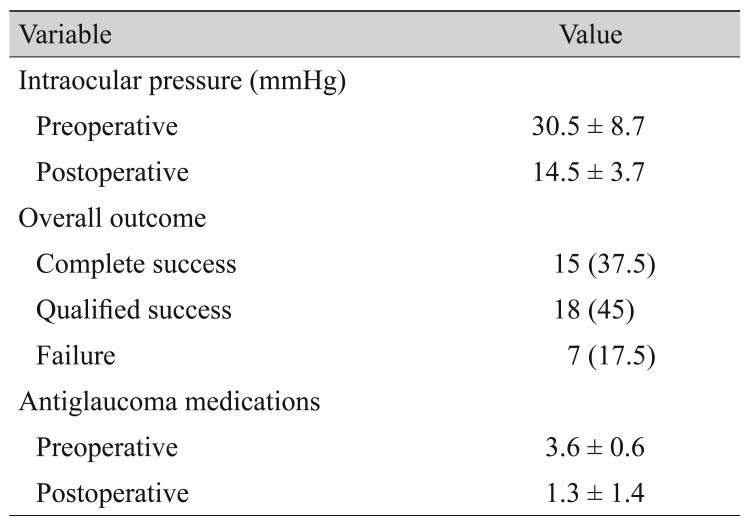
Fig. 1 shows the change in IOP after surgery. Mean IOP was lowest 1 month postoperatively, then gradually increased at 3 months and then gradually stabilized. Table 3 summarizes IOP, treatment success or failure, and number of antiglaucoma medications. Mean IOP decreased from a preoperative value of 30.5 Âą 8.7 mmHg to a postoperative value of 14.5 Âą 3.7 mmHg at the last follow-up visit (p < 0.001). The number of antiglaucoma medications also decreased from a mean of 3.6 Âą 0.6 preoperatively to 1.3 Âą 1.4 postoperatively (p < 0.001). Treatment was successful in 33 eyes (82.5%), of which 15 eyes (37.5%) met the criteria for complete success. Treatment failed in seven eyes (17.5%). The preoperative diagnoses of the seven failed cases were as follows: NVG in 3 eyes, CACG in two eyes, POAG in 1 eye, and other (secondary glaucoma due to iridocorneal endothelial syndrome) in one eye. Five eyes were primary cases, and two eyes had undergone previous glaucoma surgery. Three eyes were classified in the failure group because of loss of light perception, two eyes because of inadequate IOP control, and two eyes because of the need further glaucoma surgery. Fig. 2 shows the Kaplan-Meier survival curve for all eyes.
Table 4 shows the results of VA and SE. After surgery, VA improved in 22 eyes (55%), was maintained in eight eyes (20%), and decreased in 10 eyes (25%). In 34 eyes (excluding six eyes with intended myopic correction), mean postoperative SE was â0.53 Âą 1.14 D (range, â2.6 to +3.8 D), and 20 eyes (59%) achieved postoperative SE between â1.00 and +0.50 D. There were no significant differences in absolute refractive error among the four formulas (p = 0.562 on Friedman test).
Postoperative complications are listed in Table 5. Postoperative complications occurred in 10 eyes (25%). A hypertensive phase, defined as IOP >21 mmHg in the presence of a functioning bleb and a patent tube and occurring up to 3 months after surgery [13], was the most common complication (5 eyes, 12.5%). All cases were resolved at postoperative month 6 with antiglaucoma medication. Tube-iris touch occurred in three eyes and caused an increase in IOP in all the three eyes. Two of the three eyes underwent an additional procedure for re-adjustment of the location of the tube. Hypotony and chamber collapse occurred in two eyes; of these, they occurred in one eye after paracentesis because of high postoperative IOP (43 mmHg). Injection of viscoelastics into the anterior chamber was performed in all cases, and recovery was observed thereafter. Regular evaluation of the postoperative corneal endothelial status by specular microscopy was not performed routinely in our clinic; however, we detected one case of intermittent tube-cornea touch incidentally because of a relatively long tube. The corneal endothelial cell count decreased from 2,725/mm2 preoperatively to 1,479/mm2 at 1 year postoperative. Corneal edema was not obvious, and corneal thickness increased slightly from 551 to 565 Âľm. Tube trimming was planned for that patient.
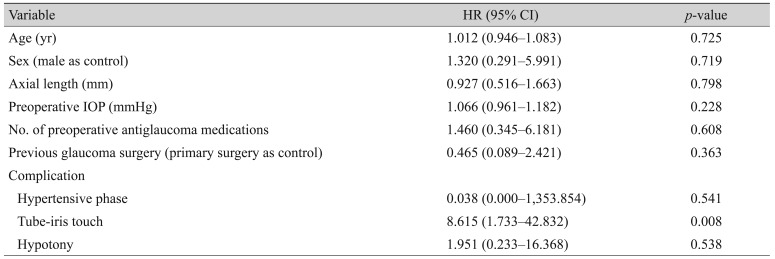
According to the Cox proportional hazards model, tip-iris touch was an independent risk factor for surgical failure (HR, 8.615; p = 0.008). Age, sex, AL, preoperative IOP, number of preoperative antiglaucoma medications, preoperative diagnosis, development of hypertensive phase, and hypotony were not significantly associated with surgical failure (p > 0.05 for all) (Table 6). Fig. 3 shows the difference in survival curves depending on whether tube-iris touch occurred (p = 0.001 by the log rank test).
Traditionally, combination cataract extraction and trabeculectomy has been performed in patients with cataract accompanied by glaucoma. However, simultaneous combined cataract extraction and trabeculectomy may not be appropriate in some situations, such as patients with a history of previous failed trabeculectomy, secondary refractory glaucoma, or previous bleb-related complications [3]. Bleb-related infections can result in devastating outcomes [16,25,26]. Conjunctival scarring from prior ocular surgeries may reduce the likelihood of successful bleb formation [27]. Furthermore, early postoperative hypotony is relatively common and has been reported in approximately 6.8% to 25.6% of cases [14,15]. In these cases, glaucoma drainage implantation can offer an alternative surgical approach to trabeculectomy for IOP control [28,29].
In the present study, 82.5% of eyes that underwent combined phacoemulsification and AGV implantation were classified as treatment success after a mean follow-up period of 18 months. IOP decreased significantly after surgery, and 55% of eyes exhibited improved VA. According to Cox proportional hazards analysis, tube-iris touch was the only risk factor for surgical failure in the present study (HR, 8.615; p = 0.008). Tube-iris touch occurred in three eyes (7.5%), two of which underwent further surgical intervention because of poor IOP control and one underwent in-office paracentesis. Two survival curves according to the presence or absence of tube-iris touch also showed a significant difference (p = 0.001 on log rank test). The exact cause of tube-iris touch remains unclear. During surgery, the depth of the anterior chamber is excessive after lens extraction due to injection of viscoelastic material in order to facilitate entry of the tube. At this time, the tube is inserted parallel to the iris plane as posteriorly as possible to avoid corneal endothelial injury. After surgery, the hyper-deepened anterior chamber may gradually become shallower. As the distance between the iris and tube shrinks, the tube tip may come into contact with and be occluded by the iris. The preoperative diagnoses of three eyes with tube-iris touch were CACG in two eyes and iridocorneal endothelial syndrome, which exhibited secondary angle closure with 360° high peripheral anterior synechiae, in one eye. Iris atrophy was observed in all three eyes. Reduced tension of the iris tissue from atrophy may result in further tube-iris touch. However, these speculations should be verified by a longitudinal study incorporating imaging data. Furthermore, despite the statistically significant association between tube-iris touch surgical failure, these results were based on only three eyes. Thus, this finding should be assessed in further large-scale studies.
Another common postoperative complication reported in GDI surgery is hypotony because of excessive early flow through the drainage implant before encapsulation around the implant occurs [30,31,32,33]. Early postoperative hypotony significantly affects surgical outcomes in patients undergoing primary phacotrabeculectomy, with an HR of 5.1 [14]. Benson et al. [34] suggested that early hypotony results in breakdown of the blood-aqueous barrier, causing the release of inflammatory mediators, which could induce enhanced bleb scarring. In contrast, in the present study, the presence of postoperative hypotony did not affect final surgical outcomes (HR, 1.951; p = 0.538). The effects of postoperative hypotony on surgical outcomes may differ in combined phacoemulsification and AGV implantation. According to Chung et al. [18], hypotony occurred in 19% of eyes that underwent combined phacoemulsification and GDI surgery. In that study, tube ligation was performed only during Baerveldt implant surgery, which accounted for about half of cases. Hoffman et al. [3] reported no postoperative hypotony after combined cataract extraction and Baerveldt GDI surgery, with tube ligation performed in all cases. The objective of tube ligation is to reduce aqueous humor flow and associated proinflammatory mediators in the initial postoperative period and to prevent early postoperative hypotony [13]. In the present study, tube ligation was performed for all cases, which resulted in a relatively low incidence of hypotony. Hypotony after combined phacoemulsification and AGV implantation tended to be temporary, and prolonged hypotony was rare [13].
A hypertensive phase was the most common complication observed in the present study, occurring in five eyes (12.5%). All cases resolved within 6 months postoperative with antiglaucoma medication. Chung et al. [18] reported an approximate 22% hypertensive phase incidence, which resolved spontaneously in all subjects with no observable effect on final IOP or VA outcomes. A previous report from our group also revealed that one-third of participants who underwent silicone plate AGV implantation experienced a hypertensive phase, which resolved in 76% of cases, and this phase was not associated with the overall success rate [35]. In the present study, we also confirmed through Cox regression analysis that the presence of a hypertensive phase is not associated with surgical failure (p = 0.541).
Recently, several randomized prospective clinical trials reported that early postoperative topical aqueous suppressant treatment (when IOP exceeds 10 mmHg) might improve AGV implantation outcomes in terms of IOP reduction, success rate, and hypertensive phase frequency [36,37]. The concentration of inflammatory mediators in tissues surrounding the AGV plate might be lower with this treatment, potentially leading to a thinner and looser Tenon's capsule and to better aqueous filtration in the long term [37]. Our study design was retrospective, and we did not randomize participants into early aqueous suppressant therapy and control groups, as done in previous studies [36,37]. Instead, we compared the mean number of postoperative topical antiglaucoma medications between eyes that exhibited a hypertensive phase and those that did not, and there were no significant differences between these two groups (2.0 Âą 0.0 vs. 1.2 Âą 1.5, respectively, p = 0.230). In our clinic, postoperative antiglaucoma medications began with a dorzolamide/timolol fixed combination, and brimonidine and prostaglandin analogs were added as needed. This topic should be explored in further prospective, randomized, comparative studies.
Several studies reported the outcomes of combined phacoemulsification and GDI surgery [3,13,17,18,38]. Hoffman et al. [3] reported a cumulative success rate of 89% at a mean follow-up duration of 15 months for combined cataract extraction and Baerveldt glaucoma drainage implantation. This as similar to our results, although the distribution of surgical indications varied somewhat: 58% of eyes in Hoffman's study had a history of failed trabeculectomy and the other 42% of eyes were undergoing primary surgery. Chung et al. [18] also reported the outcomes in 32 eyes that underwent combined phacoemulsification and Ahmed or Baerveldt implant surgery, with a cumulative success rate of 87.5% and no failures observed in the group undergoing Ahmed surgery. The mean follow-up period was 13 months, which is slightly shorter than that in the present study. In total, 47% of eyes had a history of failed trabeculectomy and 53% of eyes were primary cases. Nassiri et al. [17] reported a cumulative success rate of 87.8% 12 months postoperatively in 41 eyes that underwent combined phacoemulsification and AGV implantation. Recently, Valenzuela et al. [13] reported a cumulative success rate of 100% at a mean follow-up period of 29.5 months for combined phacoemulsification and AGV implantation. Their study included 35 eyes, 60% of which had a history of failed trabeculectomy. In the present study, treatment failure was associated with loss of light perception after surgery in three eyes, despite their IOPs being well controlled. Furthermore, 35 of 40 eyes (87.5%) achieved the criterion of success based on IOP; this finding is comparable to results reported previously [3,13,17,18]. The reported success rate of GDI surgery alone is approximately 58% to 96% [39,40,41,42,43,44,45,46]. These results suggest that concomitant phacoemulsification does not have a significant negative effect on the success rate of GDI surgery [17].
With regard to visual rehabilitation, previous studies revealed that approximately 61% to 85% of patients exhibited improved VA after surgery [3,13,18]. These results are higher than seen in the present study (55%), probably because of the relatively high proportion of subjects with NVG (22.5%) and poor preoperative VA (62.5% with less than 20 / 100) in the present study. As mentioned earlier, three eyes in our cohort were classified in the surgical failure group because of loss of light perception. They had very poor vision before surgery: light perception in two eyes and hand motion in one eye. The preoperative diagnosis was NVG from ocular ischemic syndrome in two eyes and advanced-stage POAG with previously failed trabeculectomy and AGV implantation in one eye. Optic disc pallor and relative afferent pupillary defect were obvious in all three eyes. Those eyes underwent surgery for pain control and cosmetic purposes.
Tzu et al. [38] reported the refractive outcomes of combined cataract and glaucoma surgery. About half of the eyes underwent combined trabeculectomy and the other half underwent combined glaucoma drainage device surgery. In total, 74% achieved a refractive outcome of SE between â1.00 and +0.50 D. Mean SE after combined surgery was â0.62 Âą 0.71 D, which was similar to the results of the present study (â0.53 Âą 1.14 D). This tendency was also confirmed in the case of phacotrabeculectomy. According to Ong et al. [47], combined phacotrabeculectomy resulted in a greater myopic refractive prediction error, with a postoperative SE of â0.73 Âą 0.75 D and a lower rate of satisfactory refractive outcomes than sequential phacoemulsification after trabeculectomy. In the present study, only 57% of eyes achieved a postoperative SE of â1 to +0.5 D. Notably, refractive outcomes are likely to be more unpredictable and inaccurate in cases of combined cataract and glaucoma surgery because of unstable corneal curvature, AL, and anterior chamber depth [47].
The present study has several limitations. The mean follow-up period was relatively short for assessing success or failure. In addition, postoperative corneal endothelial cell status was not assessed regularly in the present study. Corneal edema can occur because of endothelial cell injury during phacoemulsification as well as direct endothelial damage from the AGV tube [48,49,50,51]. Hoffman et al. [3] reported that 3 of 33 eyes developed corneal edema after combined surgery. We incidentally found one eye with tube-cornea touch 1 year postoperatively. Specular microscopy was performed at that time, and a significant decrease in the corneal endothelium was found. Regular follow-up of the postoperative corneal endothelium may be required in patients who have undergone combined phacoemulsification and AGV implantation.
To the best of our knowledge, this is the first study to assess risk factors for surgical failure in combined phacoemulsification and AGV implantation. Our study showed good reduction of IOP and visual rehabilitation after combined phacoemulsification and AGV implantation in patients with cataract and refractory glaucoma. In contrast to phacotrabeculectomy, hypotony did not affect surgical outcomes, which may lead to better surgical outcomes in terms of IOP control, enabling sufficient filtering of aqueous in the early postoperative period. Postoperative tube-iris touch is a significant risk factor for surgical failure that should be considered during surgery and should be carefully monitored during postoperative follow-up.
Notes
Conflict of Interest: No potential conflict of interest relevant to this article was reported.
REFERENCES
3. Hoffman KB, Feldman RM, Budenz DL, et al. Combined cataract extraction and Baerveldt glaucoma drainage implant: indications and outcomes. Ophthalmology 2002;109:1916-1920.


4. Pachimkul P, Intajak Y. Effect of lens extraction on primary angle closure in a Thai population. J Med Assoc Thai 2008;91:303-308.

5. Thomas R, Walland M, Thomas A, Mengersen K. Lowering of intraocular pressure after phacoemulsification in primary open-angle and angle-closure glaucoma: a bayesian analysis. Asia Pac J Ophthalmol (Phila) 2016;5:79-84.


6. Traverso CE. Clear-lens extraction as a treatment for primary angle closure. Lancet 2016;388:1352-1354.


7. Zhang ZM, Niu Q, Nie Y, Zhang J. Reduction of intraocular pressure and improvement of vision after cataract surgeries in angle closure glaucoma with concomitant cataract patients. Int J Clin Exp Med 2015;8:16557-16563.


8. Euswas A, Warrasak S. Intraocular pressure control following phacoemulsification in patients with chronic angle closure glaucoma. J Med Assoc Thai 2005;88(Suppl 9):S121-S125.
9. Zhuo YH, Wang M, Li Y, et al. Phacoemulsification treatment of subjects with acute primary angle closure and chronic primary angle-closure glaucoma. J Glaucoma 2009;18:646-651.


10. Tham CC, Kwong YY, Leung DY, et al. Phacoemulsification versus combined phacotrabeculectomy in medically uncontrolled chronic angle closure glaucoma with cataracts. Ophthalmology 2009;116:725-731.


11. Liu B, Guo DD, Du XJ, et al. Evaluation of Ex-PRESS implantation combined with phacoemulsification in primary angle-closure glaucoma. Medicine (Baltimore) 2016;95:e4613.



12. Wang Y, Sheng Y, Wang M, Tao J. Management of different subtypes of primary angle closure with phacoemulsification and viscogoniosynechialysis. Semin Ophthalmol 2017;32:496-500.


13. Valenzuela F, Browne A, Srur M, et al. Combined phacoemulsification and Ahmed glaucoma drainage implant surgery for patients with refractory glaucoma and cataract. J Glaucoma 2016;25:162-166.


14. Chen DZ, Koh V, Sng C, et al. Complications and outcomes of primary phacotrabeculectomy with mitomycin C in a multi-ethnic asian population. PLoS One 2015;10:e0118852.



15. Gdih GA, Yuen D, Yan P, et al. Meta-analysis of 1- versus 2-site phacotrabeculectomy. Ophthalmology 2011;118:71-76.


16. Yassin SA. Bleb-related infection revisited: a literature review. Acta Ophthalmol 2016;94:122-134.


17. Nassiri N, Nassiri N, Sadeghi Yarandi S, et al. Combined phacoemulsification and Ahmed valve glaucoma drainage implant: a retrospective case series. Eur J Ophthalmol 2008;18:191-198.


18. Chung AN, Aung T, Wang JC, Chew PT. Surgical outcomes of combined phacoemulsification and glaucoma drainage implant surgery for Asian patients with refractory glaucoma with cataract. Am J Ophthalmol 2004;137:294-300.


19. Coleman AL, Mondino BJ, Wilson MR, Casey R. Clinical experience with the Ahmed glaucoma valve implant in eyes with prior or concurrent penetrating keratoplasties. Am J Ophthalmol 1997;123:54-61.


20. Kee C. Prevention of early postoperative hypotony by partial ligation of silicone tube in Ahmed glaucoma valve implantation. J Glaucoma 2001;10:466-469.


21. Krupin T, Ritch R, Camras CB, et al. A long Krupin-Denver valve implant attached to a 180 degrees scleral explant for glaucoma surgery. Ophthalmology 1988;95:1174-1180.


22. Canto AP, Chhadva P, Cabot F, et al. Comparison of IOL power calculation methods and intraoperative wavefront aberrometer in eyes after refractive surgery. J Refract Surg 2013;29:484-489.


23. Fram NR, Masket S, Wang L. Comparison of intraoperative aberrometry, OCT-based IOL formula, Haigis-L, and Masket formulae for IOL power calculation after laser vision correction. Ophthalmology 2015;122:1096-1101.


24. Nihalani BR, VanderVeen DK. Comparison of intraocular lens power calculation formulae in pediatric eyes. Ophthalmology 2010;117:1493-1499.


25. Waheed S, Ritterband DC, Greenfield DS, et al. Bleb-related ocular infection in children after trabeculectomy with mitomycin C. Ophthalmology 1997;104:2117-2120.


26. Yamamoto T, Kuwayama Y, Nomura E, et al. Changes in visual acuity and intra-ocular pressure following bleb-related infection: the Japan Glaucoma Society Survey of Bleb-related Infection Report 2. Acta Ophthalmol 2013;91:e420-e426.


27. Rockwood EJ, Parrish RK 2nd, Heuer DK, et al. Glaucoma filtering surgery with 5-fluorouracil. Ophthalmology 1987;94:1071-1078.


28. Englert JA, Freedman SF, Cox TA. The Ahmed valve in refractory pediatric glaucoma. Am J Ophthalmol 1999;127:34-42.


29. Gedde SJ, Panarelli JF, Banitt MR, Lee RK. Evidenced-based comparison of aqueous shunts. Curr Opin Ophthalmol 2013;24:87-95.


30. Fellenbaum PS, Almeida AR, Minckler DS, et al. Krupin disk implantation for complicated glaucomas. Ophthalmology 1994;101:1178-1182.


31. Lotufo DG. Postoperative complications and visual loss following Molteno implantation. Ophthalmic Surg 1991;22:650-656.


32. Melamed S, Cahane M, Gutman I, Blumenthal M. Postoperative complications after Molteno implant surgery. Am J Ophthalmol 1991;111:319-322.


33. Minckler DS, Heuer DK, Hasty B, et al. Clinical experience with the single-plate Molteno implant in complicated glaucomas. Ophthalmology 1988;95:1181-1188.


34. Benson SE, Mandal K, Bunce CV, Fraser SG. Is post-trabeculectomy hypotony a risk factor for subsequent failure? A case control study. BMC Ophthalmol 2005;5:7



35. Won HJ, Sung KR. Hypertensive phase following silicone plate Ahmed glaucoma valve implantation. J Glaucoma 2016;25:e313-e317.


36. Law SK, Kornmann HL, Giaconi JA, et al. Early aqueous suppressant therapy on hypertensive phase following glaucoma drainage device procedure: a randomized prospective trial. J Glaucoma 2016;25:248-257.


37. Pakravan M, Rad SS, Yazdani S, et al. Effect of early treatment with aqueous suppressants on Ahmed glaucoma valve implantation outcomes. Ophthalmology 2014;121:1693-1698.


38. Tzu JH, Shah CT, Galor A, et al. Refractive outcomes of combined cataract and glaucoma surgery. J Glaucoma 2015;24:161-164.


39. Burgoyne JK, WuDunn D, Lakhani V, Cantor LB. Outcomes of sequential tube shunts in complicated glaucoma. Ophthalmology 2000;107:309-314.


40. Coleman AL, Hill R, Wilson MR, et al. Initial clinical experience with the Ahmed glaucoma valve implant. Am J Ophthalmol 1995;120:23-31.


41. Gedde SJ, Schiffman JC, Feuer WJ, et al. Treatment outcomes in the tube versus trabeculectomy study after one year of follow-up. Am J Ophthalmol 2007;143:9-22.


42. Huang MC, Netland PA, Coleman AL, et al. Intermediate-term clinical experience with the Ahmed glaucoma valve implant. Am J Ophthalmol 1999;127:27-33.


43. Ozdal PC, Vianna RN, Deschenes J. Ahmed valve implantation in glaucoma secondary to chronic uveitis. Eye (Lond) 2006;20:178-183.


44. Taglia DP, Perkins TW, Gangnon R, et al. Comparison of the Ahmed glaucoma valve, the Krupin eye valve with disk, and the double-plate Molteno implant. J Glaucoma 2002;11:347-353.


45. Wilson MR, Mendis U, Paliwal A, Haynatzka V. Long-term follow-up of primary glaucoma surgery with Ahmed glaucoma valve implant versus trabeculectomy. Am J Ophthalmol 2003;136:464-470.


46. Wilson MR, Mendis U, Smith SD, Paliwal A. Ahmed glaucoma valve implant vs trabeculectomy in the surgical treatment of glaucoma: a randomized clinical trial. Am J Ophthalmol 2000;130:267-273.


47. Ong C, Nongpiur M, Peter L, Perera SA. Combined approach to phacoemulsification and trabeculectomy results in less ideal refractive outcomes compared with the sequential approach. J Glaucoma 2016;25:e873-e878.


48. Hayashi K, Hayashi H, Nakao F, Hayashi F. Risk factors for corneal endothelial injury during phacoemulsification. J Cataract Refract Surg 1996;22:1079-1084.


49. Kohlhaas M, Klemm M, Kammann J, Richard G. Endothelial cell loss secondary to two different phacoemulsification techniques. Ophthalmic Surg Lasers 1998;29:890-895.


Fig. 1
Graph showing trends in mean intraocular pressure from preoperative to the last follow-up visit (error bars indicate 95% confidence intervals).
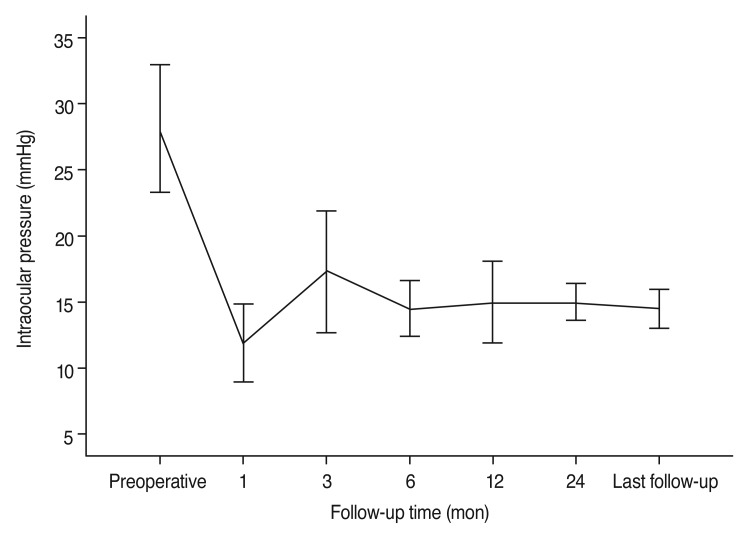
Fig. 2
Kaplan-Meier survival curve with 95% confidence intervals for the total number of eyes undergoing combined phacoemulsification and Ahmed valve implantation.
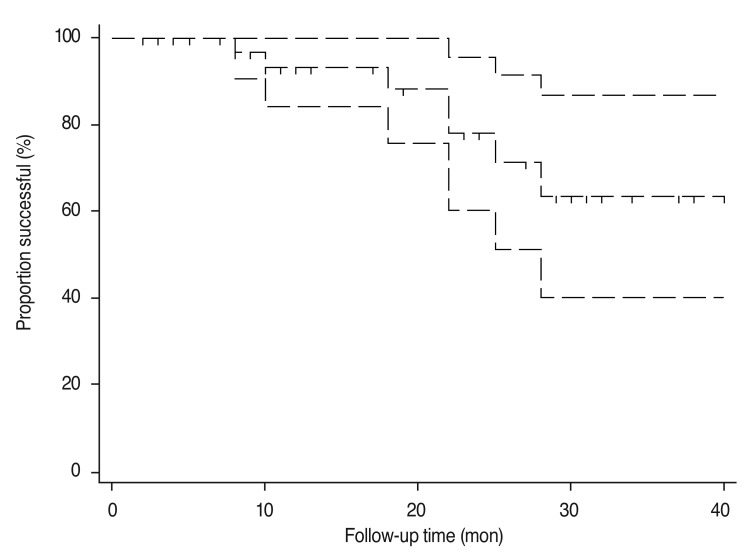
Table 2
Preoperative diagnosis for eyes that underwent combined phacoemulsification and Ahmed glaucoma valve implantation

Table 4
Visual acuity, postoperative spherical equivalent, and comparison of refractive errors among various intraocular lens power calculation formulas

- TOOLS
-
METRICS

-
- 5 Crossref
- 0 Scopus
- 2,672 View
- 32 Download
- Related articles





 PDF Links
PDF Links PubReader
PubReader Full text via DOI
Full text via DOI Full text via PMC
Full text via PMC Download Citation
Download Citation Print
Print



