 |
 |
| Korean J Ophthalmol > Volume 32(2); 2018 > Article |
Abstract
Methods
A retrospective chart review was conducted. Charts of patients with idiopathic macular holes who underwent surgery by a single surgeon at Severance Hospital between January 1, 2013 and July 31, 2015 were reviewed. The best-corrected visual acuity (BCVA) score was recorded preoperatively and at 1 day and 1, 3, 6, 9, and 12 months after surgery. The variables of age, sex, macular hole size, basal hole diameter, choroidal thickness, and axial length were also noted.
Results
Twenty-six eyes of 26 patients were evaluated. Twenty-five patients (96.2%) showed successful macular hole closure after the primary operation. The BCVA stabilized 6 months postoperatively. A large basal hole diameter (p = 0.006) and thin choroid (p = 0.005) were related to poor visual outcomes. Poor preoperative BCVA (p < 0.001) and a thick choroid (p = 0.020) were associated with greater improvement in BCVA after surgery.
Optical coherence tomography (OCT) technology has led to a deeper understanding of the pathophysiology of macular holes (MHs) [1]. With advancements in surgical techniques, from gas tamponade to internal limiting membrane staining with indocyanine green dye, the primary success rates of MH surgeries have steadily increased [2,3,4]. Some efforts have been made to determine the prognostic factors of favorable MH surgery outcomes [5,6,7]. To date, the size and stage of the MH, duration of symptoms, and preoperative visual acuity have been reported as preoperative factors [5]; however, recovery of the macular contour, the external limiting membrane (ELM), and the ellipsoid zone are known to affect recovery of vision after surgery [6,7].
It has been reported that MH patients have a thin choroid and decreased choroidal perfusion [8,9], but it remains unknown whether choroidal thickness or perfusion affect the surgical outcome. Additionally, there have been few reports about the time required for visual recovery after MH repair. Thus, in this study, we investigated the time to visual recovery after MH surgery and the related prognostic factors, including choroidal thickness.
The institutional review board of Yonsei University College of Medicine approved this study (4-2016-1033), which adhered to the tenets of the Declaration of Helsinki. All patients provided written informed consent before any procedure was performed. A retrospective chart review was performed for all patients with idiopathic MHs, who underwent surgery by a single surgeon (SSK) at Severance Hospital, Seoul, Korea, between January 1, 2013 and July 31, 2015. Patients were excluded if they were lost to follow-up within one year after surgery, developed a secondary MH, had an axial length exceeding 26.5 mm, or underwent previous vitrectomy.
All patients underwent a thorough ophthalmic evaluation prior to surgery, including assessment of the best-corrected visual acuity (BCVA) and a fundus examination (Spectralis OCT; Heidelberg Engineering, Heidelberg, Germany). Patients with a full thickness MH were advised to undergo surgery. Surgery was performed using a standard 3-port, 23-gauge pars plana vitrectomy. When a cataract was present, a combined phacovitrectomy was performed. To remove tangential traction, epiretinal membranectomy was performed when a premacular membrane was present. When there was no visible premacular membrane, the internal limiting membrane was peeled with indocyanine green staining. After removing tangential traction around the MH, air-fluid exchange was performed, followed by 10% to 14% C3F8 tamponade. Patients were instructed to remain in a prone position for about one week after the surgery.
Patient age, sex, and involved eye were noted, and the BCVA was measured before surgery and at 1 day and 1, 3, 6, 9, and 12 months after the operation. Size of the MH, basal hole diameter, and subfoveal choroidal thickness were measured using the caliper function in the Heidelberg software (Fig. 1). The basal hole diameter of the MH was measured as the linear length of retinal detachment. Subfoveal choroidal thickness was measured three times by a single observer (KSH) with enhanced depth imaging, using a method previously described by Boonarpha et al. [10], as the distance from the hyper-reflective line under the retinal pigment epithelium to the choroid-sclera interface. Recovery of the ELM and the ellipsoid zone was monitored with serial OCT scans during follow-up. Axial length was measured preoperatively with the IOL-Master 500 (Carl Zeiss, Dublin, CA, USA).
Changes in BCVA were analyzed by repeated measures analysis of variance. Multivariate linear regression was performed to determine prognostic factors after MH repair. Fisher's exact test and the Mann-Whitney U-test were used to compare characteristics and outcomes between the ELM and ellipsoid zone recovery. Reliability analysis was used to assess the reproducibility of subfoveal choroidal thickness measurements. Statistical analysis was performed using IBM SPSS ver. 21.0 (IBM Corp., Armonk, NY, USA). A p-value less than 0.05 was considered statistically significant.
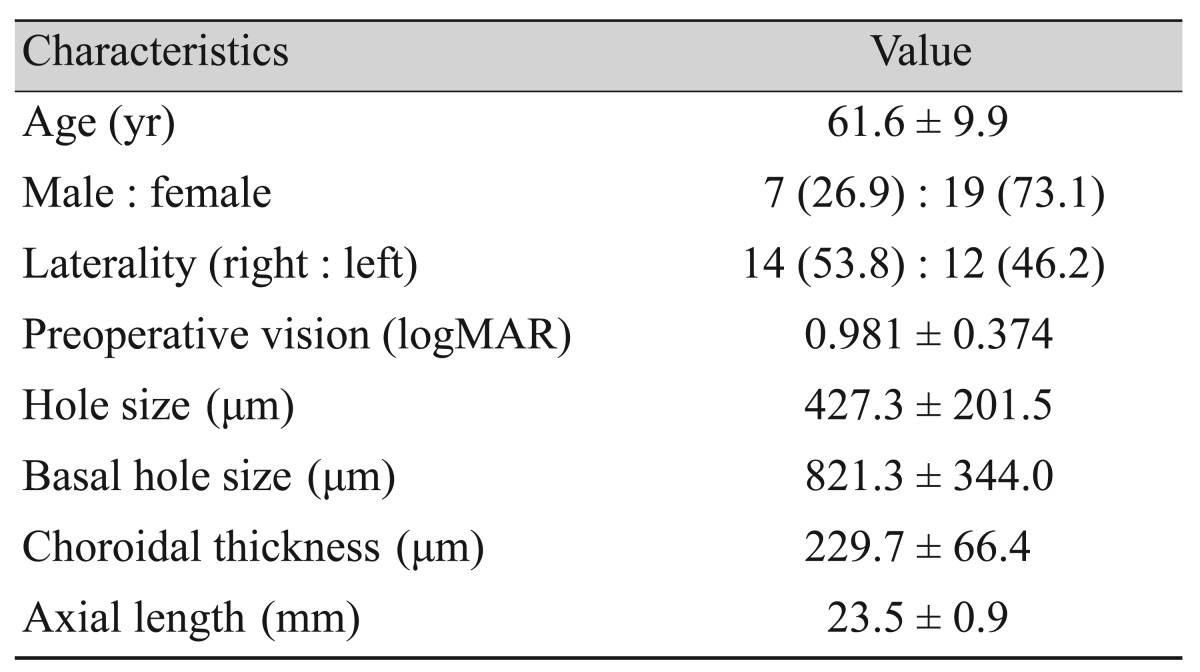
Twenty-six eyes of 26 patients were included in this study. Demographic factors and initial findings are listed in Table 1. The mean age of the patients was 61.6 years, and 73.1% (19 patients) were female. The mean BCVA was 0.981 in logarithm of the minimum angle of resolution scale prior to the operation. Visual acuity decreased to a mean of 3.077 1 day after the operation, due to gas filling. All 26 patients completed all routine follow-up visits. The mean BCVA values at 1, 3, 6, 9, and 12 months were 0.797, 0.592, 0.495, 0.496, and 0.427, respectively. When these BCVA values were compared with the final (12-month postoperative) BCVA, preoperative vision and 1-day postoperative vision were found to be significantly different (p < 0.001). Differences in BCVA were still significant at 1 and 3 months after the operation (1 month, p = 0.034; 3 months, p = 0.040). BCVA did not show improvements beyond 6 months after surgery (6 months, p = 1.000; 9 months, p = 1.000) (Fig. 2).
Measurement of subfoveal choroidal thickness showed good reliability, with an intraclass correlation coefficient of 0.992 (p < 0.001). Postoperative choroidal thickness measured at 3, 6, 9, and 12 months was 227.3 (n = 22), 218.7 (n = 26), 214.2 (n = 25), and 223.6 (n = 23), respectively. Thinning of the choroid was noted in all postoperative thickness measurements compared to the preoperative choroidal thickness measurement (3 months, p = 0.001; 6 months, p = 0.004; 9 months, p < 0.001; 12 months, p = 0.001). Changes in choroidal thickness did not correlate with preoperative BCVA (p = 0.427), 12-month postoperative BCVA (p = 0.543), preoperative choroidal thickness (p = 0.935), 12-month postoperative choroidal thickness (p = 0.394), or preoperative axial length (p = 0.687).
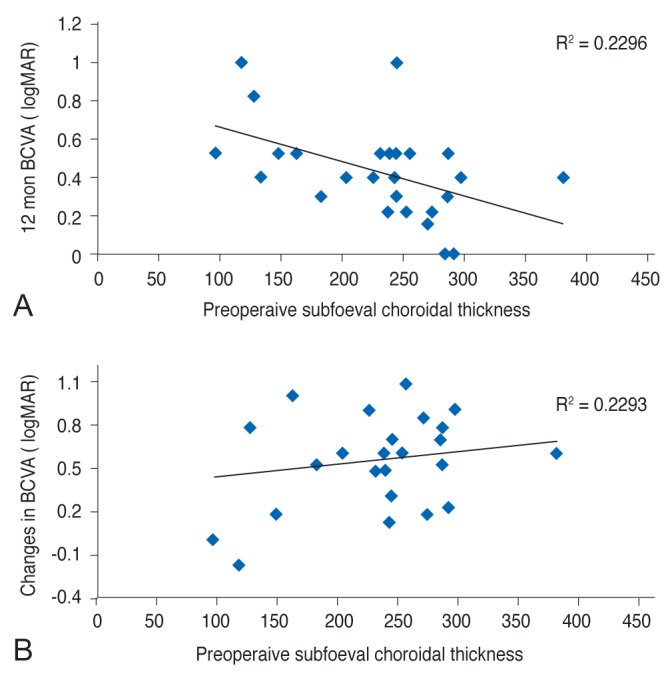
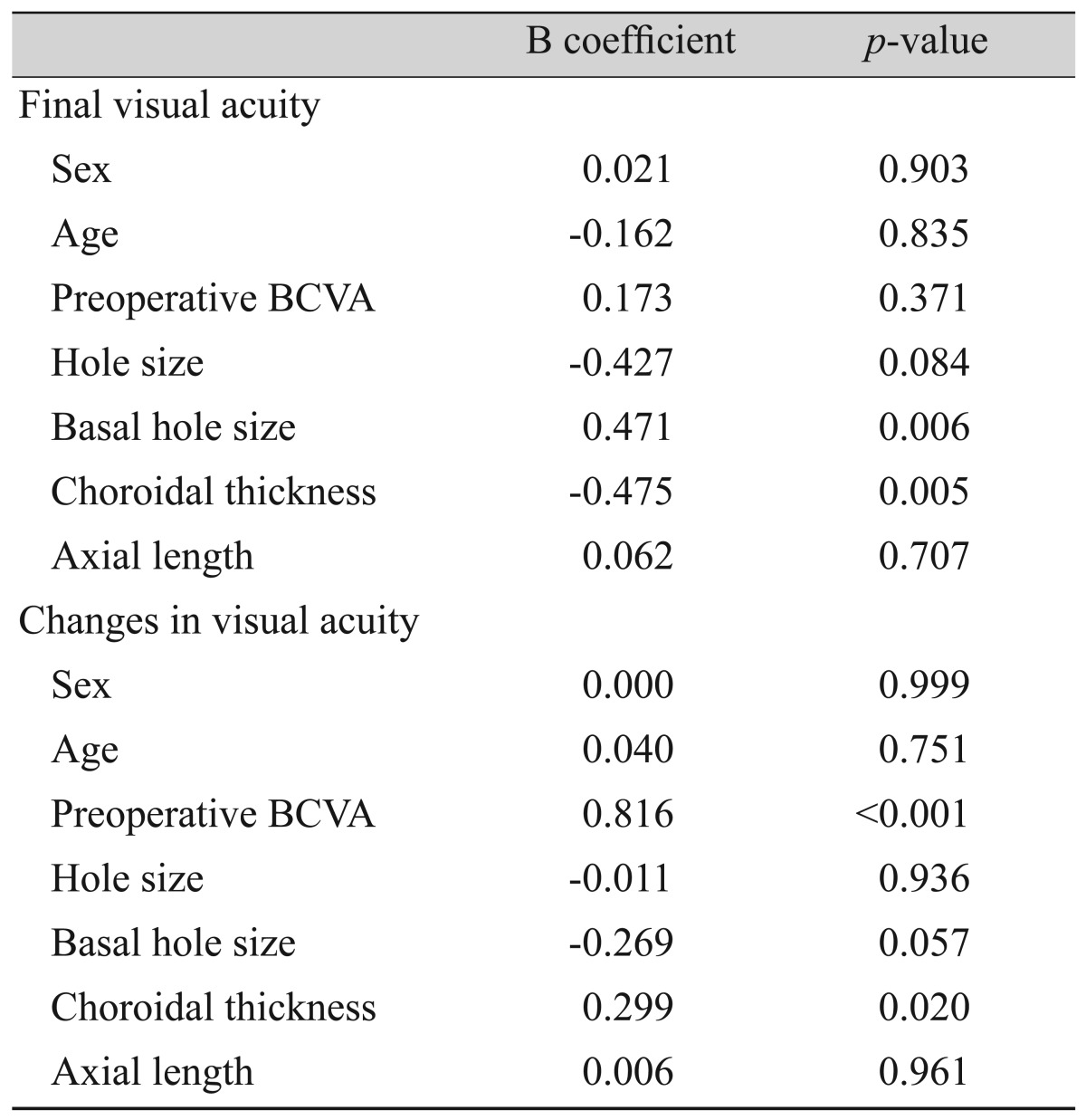
To evaluate the factors affecting final BCVA, the variables of sex, age, preoperative BCVA, MH size, basal hole diameter, choroidal thickness, and axial length were evaluated using multivariate linear regression analysis. This revealed that the final visual acuity was affected by basal hole diameter and choroidal thickness (R2 = 0.450, p = 0.001). A larger basal hole diameter was associated with a poor final BCVA (95% confidence interval [CI], 0.000 to 0.001). A greater preoperative choroidal thickness provided a protective effect, as patients with a thick choroid had a better final BCVA (95% CI, 0.000 to 0.001). When these factors were evaluated against changes in BCVA, preoperative BCVA and choroidal thickness were significantly associated (R2 = 0.680, p < 0.001). Patients with poor preoperative BCVA showed greater visual recovery (95% CI, 0.521 to 0.974). Choroidal thickness also had a positive relationship with visual acuity changes (95% CI, 0.000 to 0.003) (Table 2 and Fig. 3A, 3B).
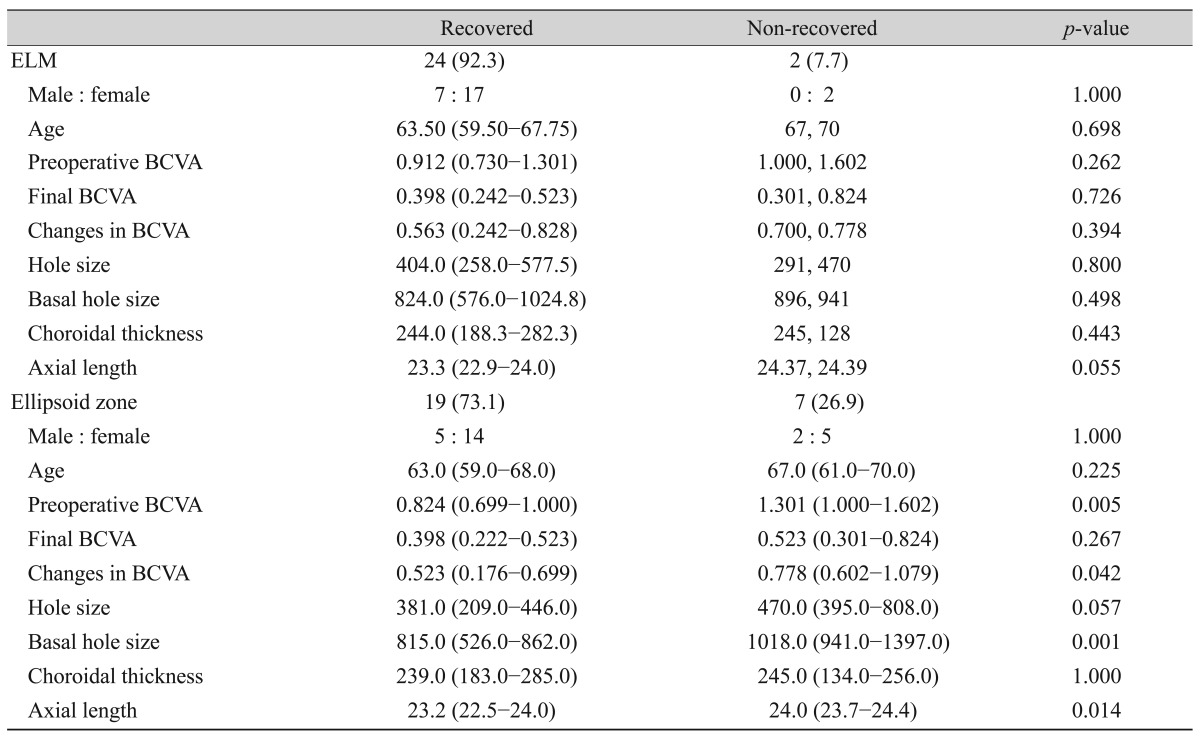
Twenty-four (92.3%) patients showed ELM recovery at a median of 1 month (1st to 3rd quartile, 1 to 4.50 months) after surgery. No factors were significantly related to a difference in ELM recovery. The ellipsoid zone recovered in 19 (73.1%) patients, by a median time of 3 months (1st to 3rd quartile, 1 to 8.0 months) after the operation. Patients with a recovered ellipsoid zone had better preoperative BCVA, smaller basal hole diameter, and shorter axial length. Although the final BCVA was better in the ellipsoid zone-recovered group, the difference was not statistically significant (Table 3).
In this study, we investigated the time to visual recovery after MH surgery and the related prognostic factors, including choroidal thickness. Previously, the closure rate after MH surgery was 58%, but advancements in surgical techniques and devices have led to an improvement in the closure rate to 89% to 100% [4,11,12]. In this study, we were able to achieve closure in 25 (96.2%) of 26 cases, using a standard 23-gauge vitrectomy system. The single patient with incomplete closure had a 395-┬Ąm sized MH, with a 1,397-┬Ąm basal hole diameter. He underwent a second operation 1 month after the first, during which the MH was successfully closed.
Shinoda et al. [13] reported visual recovery at 3 months after MH surgery in a 25-gauge vitrectomy group and at 9 months after surgery in a 20-gauge vitrectomy group. Less trauma to the ocular surface, a shorter operation time, and a reduced amount of irrigation fluid were considered to be reasons for the shorter time to visual recovery in the 25-gauge vitrectomy group. In this study, we performed 23-gauge vitrectomy, and vision stabilized by 6 months after the operation. Leonard et al. [14] and Purtskhvanidze et al. [15] reported continuous visual improvement after MH surgery after following patients for up to 96 months. We observed continuous elevation in visual acuity for 6 months after the operation, and recovery of ELM and the ellipsoid zone was still observed at 35 and 38 months after surgery. However, changes in visual acuity were not statistically significant.
In 1986, Morgan and Schatz [16] hypothesized that the first step in the development of a MH is choroidal vascular change. They suggested that the majority of patients with an MH had cardiovascular disease, and decreased subfoveal choroidal circulation was noticed during fluorescein angiography. Allen et al. [17] induced MHs in monkeys with an Nd-YAG laser, which resulted in a choriocapillaris perfusion disorder. Histologically, the choriocapillaris was replaced with fibroblasts and connective tissue. Other reports have also suggested that choroidal thickness is decreased in idiopathic MH patients compared to a control group when measured using OCT [8,9]. Aras et al. [18] have reported that patients with stage 4 and stage 1a MHs had reduced subfoveal choroid circulation compared to controls, as determined using a scanning laser Doppler flowmeter. It is suspected that decreased choroidal circulation and a thinner choroid can cause macular hypoperfusion, and that deficits in several protective factors leave the macula vulnerable to various types of damage [16,18]. In this study, choroidal thickness was not only related to the development of an MH, but final visual acuity and visual acuity change were also closely related. Choroidal thickness showed a protective effect on BCVA and on the degree of visual improvement obtained. We hypothesize that a thinner choroid can cause additional decreases in choroidal circulation during compression by gas tamponade and can affect MH healing. If so, prolonged prone positioning of patients after surgery might have a hazardous effect on choroidal circulation. Further studies on the changes in choroidal circulation during the tamponade period are needed.
Choroidal thickness has been reported to decrease with age and with longer axial length [19,20]. It is also known that there is a diurnal variation of approximately 30 ┬Ąm in choroidal thickness, and that the thickness is also affected by changes in blood pressure and drugs, such as sildenafil and steroids [21,22,23]. In this study, patients with an axial length exceeding 26.5 mm were excluded. However, further investigation of the relationship between choroidal thickness and visual prognosis in myopic MH patients is necessary. Choroidal thickness and choroidal circulation are not synonymous. Sogawa et al. [24] reported that choroidal blood flow was not correlated with choroidal thickness, as measured by laser Doppler, but their study subjects were young, healthy, and few in number.
ELM and ellipsoid zone recovery also affect visual recovery after MH surgery [6,7]. We compared multiple factors of the ELM and ellipsoid zone-recovered groups with those of the unrecovered group, but we found no factors affecting ELM recovery. Given that only two patients (7.7%) had unrecovered ELM, the small sample size might underlie the lack of statistically significant differences. In the case of ellipsoid zone recovery, patients with a larger basal hole diameter, poor initial visual acuity, and greater axial length had a poor recovery rate. The change in visual acuity was found to be greater in the group without recovery of the ellipsoid zone, but this was due to the differences in preoperative visual acuity. Landa et al. [25] asserted that recovery of ELM is essential for ellipsoid zone recovery. We also analyzed OCT data and found that ellipsoid zone recovery was seen only after the ELM was restored.
There are several limitations to this study. First, we did not measure the choroidal circulation, and further investigation is needed to determine whether the choroidal circulation actually affects prognosis. If so, it will be necessary to confirm whether the postoperative course is different in cases where the choroidal circulation is increased. Secondly, because this was a single center study of only Korean patients, the results might not be generalizable to different ethnicities. In addition, there are many patients who were not assessed for the duration of symptoms; thus, future studies will need to consider various parameters, including duration of symptoms and choroidal thickness.
In conclusion, visual acuity stabilized after 6 months in MH patients who underwent 23-gauge vitrectomy. Final visual acuity was poor in patients with a larger basal hole diameter. Greater choroidal thickness was a protective prognostic factor for final BCVA. Greater visual improvement was seen in patients with a poor initial BCVA and thick subfoveal choroid. The ellipsoid zone showed poor recovery in patients with a large basal hole diameter and a greater axial length. Based on these results, we were able to identify the factors that influence visual recovery after MH surgery. This information will allow for better stratification of patients who are more likely to benefit from surgical MH repair.
Notes
This study was presented as e-poster at Retina World Congress on February 25, 2017 in Fort Lauderdale, FL, USA.
Notes
Conflict of Interest: No potential conflict of interest relevant to this article was reported.
REFERENCES
1. Gaudric A, Haouchine B, Massin P, et al. Macular hole formation: new data provided by optical coherence tomography. Arch Ophthalmol 1999;117:744-751.


2. Haritoglou C, Gandorfer A, Gass CA, et al. Indocyanine green-assisted peeling of the internal limiting membrane in macular hole surgery affects visual outcome: a clinicopathologic correlation. Am J Ophthalmol 2002;134:836-841.


3. Brooks HL Jr. Macular hole surgery with and without internal limiting membrane peeling. Ophthalmology 2000;107:1939-1948.


4. Vaziri K, Schwartz SG, Kishor KS, et al. Rates of reoperation and retinal detachment after macular hole surgery. Ophthalmology 2016;123:26-31.


5. Ullrich S, Haritoglou C, Gass C, et al. Macular hole size as a prognostic factor in macular hole surgery. Br J Ophthalmol 2002;86:390-393.



6. Sano M, Shimoda Y, Hashimoto H, Kishi S. Restored photoreceptor outer segment and visual recovery after macular hole closure. Am J Ophthalmol 2009;147:313-318.


7. Itoh Y, Inoue M, Rii T, et al. Correlation between length of foveal cone outer segment tips line defect and visual acuity after macular hole closure. Ophthalmology 2012;119:1438-1446.


8. Reibaldi M, Boscia F, Avitabile T, et al. Enhanced depth imaging optical coherence tomography of the choroid in idiopathic macular hole: a cross-sectional prospective study. Am J Ophthalmol 2011;151:112-117.


9. Zeng J, Li J, Liu R, et al. Choroidal thickness in both eyes of patients with unilateral idiopathic macular hole. Ophthalmology 2012;119:2328-2333.


10. Boonarpha N, Zheng Y, Stangos AN, et al. Standardization of choroidal thickness measurements using enhanced depth imaging optical coherence tomography. Int J Ophthalmol 2015;8:484-491.


11. Kelly NE, Wendel RT. Vitreous surgery for idiopathic macular holes: results of a pilot study. Arch Ophthalmol 1991;109:654-659.


12. Briand S, Chalifoux E, Tourville E, et al. Prospective randomized trial: outcomes of SF6 versus C3F8 in macular hole surgery. Can J Ophthalmol 2015;50:95-100.


13. Shinoda H, Shinoda K, Satofuka S, et al. Visual recovery after vitrectomy for macular hole using 25-gauge instruments. Acta Ophthalmol 2008;86:151-155.


14. Leonard RE 2nd, Smiddy WE, Flynn HW Jr, Feuer W. Long-term visual outcomes in patients with successful macular hole surgery. Ophthalmology 1997;104:1648-1652.


15. Purtskhvanidze K, Treumer F, Junge O, et al. The long-term course of functional and anatomical recovery after macular hole surgery. Invest Ophthalmol Vis Sci 2013;54:4882-4891.


16. Morgan CM, Schatz H. Involutional macular thinning: a pre-macular hole condition. Ophthalmology 1986;93:153-161.


17. Allen RD, Brown J Jr, Zwick H, et al. Laser-induced macular holes demonstrate impaired choroidal perfusion. Retina 2004;24:92-97.


18. Aras C, Ocakoglu O, Akova N. Foveolar choroidal blood flow in idiopathic macular hole. Int Ophthalmol 2004;25:225-231.


19. Wei WB, Xu L, Jonas JB, et al. Subfoveal choroidal thickness: the Beijing Eye Study. Ophthalmology 2013;120:175-180.


20. Tan CS, Ouyang Y, Ruiz H, Sadda SR. Diurnal variation of choroidal thickness in normal, healthy subjects measured by spectral domain optical coherence tomography. Invest Ophthalmol Vis Sci 2012;53:261-266.


21. Chakraborty R, Read SA, Collins MJ. Diurnal variations in axial length, choroidal thickness, intraocular pressure, and ocular biometrics. Invest Ophthalmol Vis Sci 2011;52:5121-5129.


22. Sonoda S, Sakamoto T, Yamashita T, et al. Effect of intravitreal triamcinolone acetonide or bevacizumab on choroidal thickness in eyes with diabetic macular edema. Invest Ophthalmol Vis Sci 2014;55:3979-3985.


23. Vance SK, Imamura Y, Freund KB. The effects of sildenafil citrate on choroidal thickness as determined by enhanced depth imaging optical coherence tomography. Retina 2011;31:332-335.


Fig.┬Ā1
Macular hole parameters measured in optical coherence tomography scans. a = hole size; b = basal hole size; c = choroidal thickness.
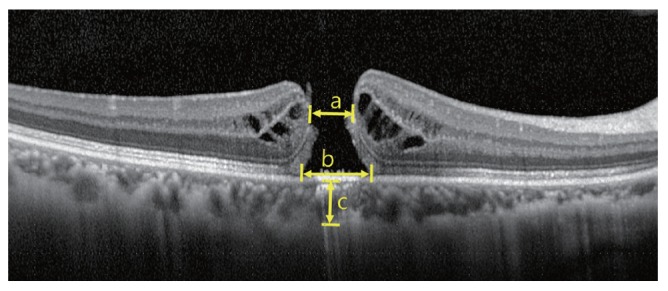
Fig.┬Ā2
Changes in best-corrected visual acuity (BCVA) after macular hole repair. logMAR = logarithm of the minimum angle of resolution. *p < 0.05, **p < 0.001.
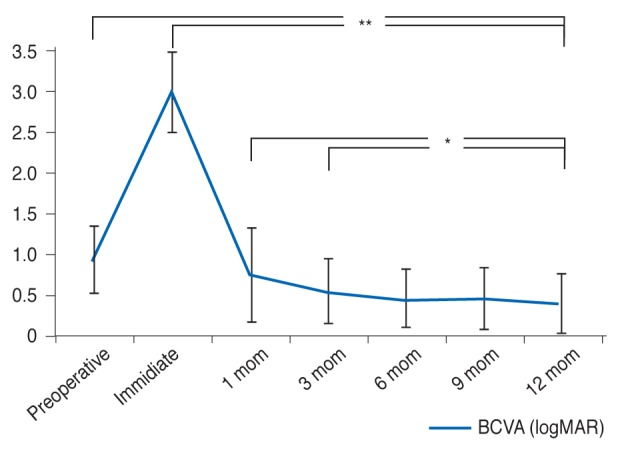
Fig.┬Ā3
Analysis of correlation of choroidal thickness with (A) final best-corrected visual acuity (BCVA) and (B) changes in BCVA. logMAR = logarithm of the minimum angle of resolution.
- TOOLS
-
METRICS

-
- 15 Crossref
- 0 Scopus
- 3,671 View
- 84 Download
- Related articles
-
Dry Eye After Cataract Surgery and Associated Intraoperative Risk Factors2009 June;23(2)



 PDF Links
PDF Links PubReader
PubReader Full text via DOI
Full text via DOI Full text via PMC
Full text via PMC Download Citation
Download Citation Print
Print



