 |
 |
| Korean J Ophthalmol > Volume 31(4); 2017 > Article |
Abstract
Purpose
The purpose of this study is to investigate new prognostic factors in associated with primary anatomical failure after scleral buckling (SB) for uncomplicated rhegmatogenous retinal detachment (RRD).
Methods
The medical records of patients with uncomplicated RRD treated with SB were retrospectively reviewed. Eyes with known prognostic factors for RRD, such as fovea-on, proliferative vitreoretinopathy, pseudophakia, aphakia, multiple breaks, or media opacity, were excluded. Analysis was performed to find correlations between anatomical success and various parameters, including age.
Results
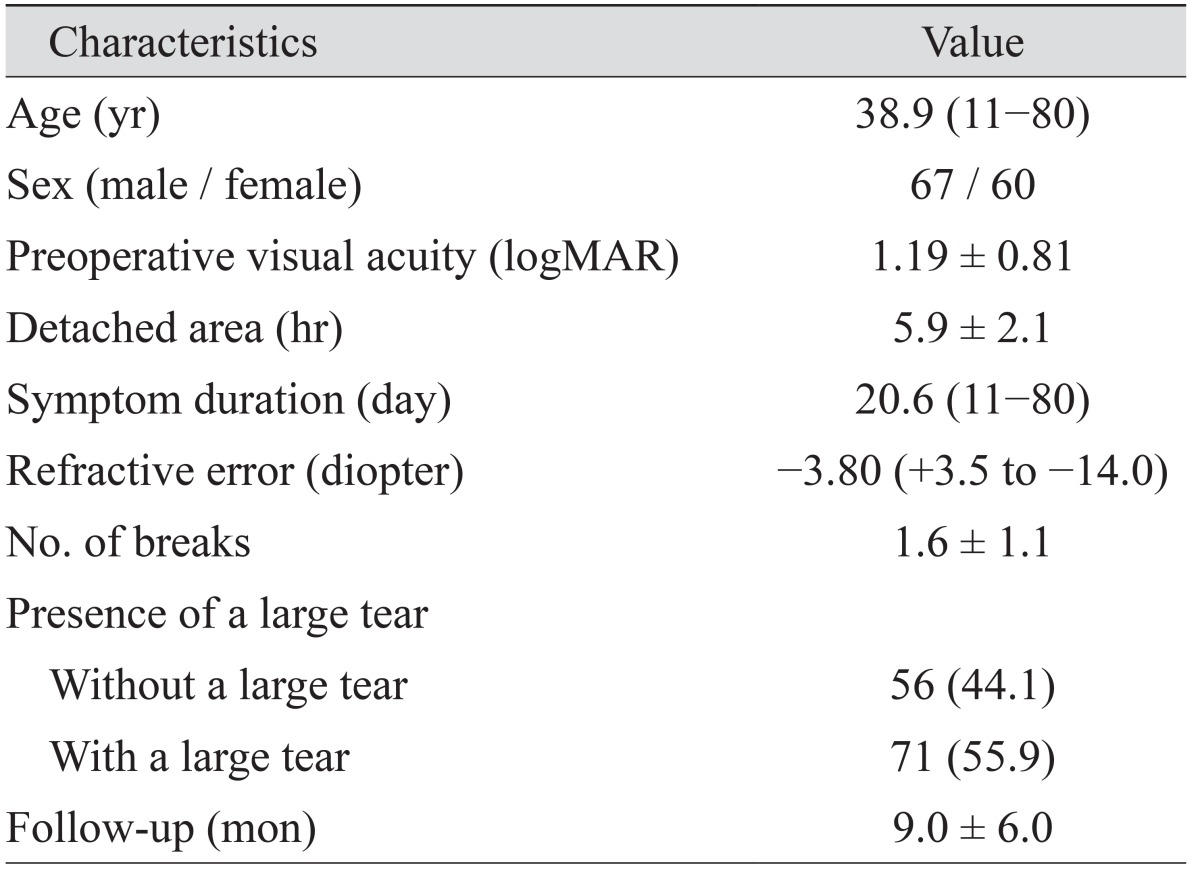
This study analyzed 127 eyes. Binary logistic regression analysis revealed that older age (≥35) was the sole independent prognostic factor (odds ratio, 3.5; p = 0.022). Older age was correlated with worse preoperative visual acuity (p < 0.001), shorter symptom duration (p < 0.001), presence of a large tear (p < 0.001), subretinal fluid drainage (p < 0.001), postoperative macular complications (p = 0.048), and greater visual improvement (p = 0.003).
Conclusions
Older age (≥35) was an independent prognostic factor for primary anatomical failure in SB for uncomplicated RRD. The distinguished features of RRD between older and younger patients suggest that vitreous liquefaction and posterior vitreous detachment are important features associated with variation in surgical outcomes.
Rhegmatogenous retinal detachment (RRD) is a vision-threatening disease in which the macula is damaged irreversibly by being detached from the retinal pigment epithelium. Scleral buckling (SB) was previously the only standard treatment for RRD for the past several decades. Previous research has identified several prognostic factors associated with anatomical success after SB [1,2,3,4,5]. The identified preoperative factors include proliferative vitreoretinopathy [1,2], pseudophakic [3,4], media opacity [1], and multiple retinal breaks [1,2,5], also known as complicated RRD; pars plana vitrectomy (PPV) is advocated for these cases.
Vitrectomy is becoming a popular technique for treating RRD, while many surgeons empirically use SB as a primary procedure in young phakic RRD [1,6]. However, there is no evidence or guidelines for these choices in uncomplicated phakic RRD.
The current study was conducted to investigate the prognostic factors after SB in uncomplicated phakic RRD, which excluded known prognostic factors.
The study was approved by the ethical committee of Pusan National University in accordance with the rules set forth in the Helsinki Declaration. We retrospectively reviewed the data for consecutive patients diagnosed with uncomplicated fovea-involved RRD in a phakic eye treated with SB and who were followed up for 3 months or more between January 2011 and December 2012. All patients agreed to treatment with SB and provided written informed consent.
Cases with an attached fovea; history of any intraocular surgery, including phacoemulsification, obvious vitreous opacity, or hemorrhage, multiple breaks (four or more); or other ocular diseases affecting visual function were excluded. Cases with proliferative vitreoretinopathy of grade C were also excluded.
The preoperative parameters consisted of age, visual acuity (VA), detachment area, symptom duration, refractive error, and presence of a large tear. Detachment area was measured as clock hours at the equator. Presence of a large tear was defined as a tear with the largest dimension of 0.5 disc diameters or more. Our database indicated that 35 years of age was an inflection point, therefore patients were sorted into two groups (<35 and ≥35).
Intraoperative characteristics were evaluated, including subretinal fluid (SRF) drainage and surgeon experience. A surgeon was classified as experienced if they had surgically treated SB for 5 years or more.
Surgical outcomes, such as anatomical success, postoperative VA, visual improvement, and optical coherent tomography findings, were assessed 3 months after SB. Primary anatomical success was defined as a retina that remained attached for 3 months or more after the first surgery. A small amount of localized f luid in the macula or periphery, which decreased during follow-up, was not considered surgical failure. Macular complications referred to a macular hole or epiretinal membrane that was significant enough to require surgical intervention. Final visual outcome was divided into two groups, the logarithm of minimum angle resolution (logMAR) score higher or lower than 0.2, as our database indicated that 0.2 was the median value.
The primary outcome of this study was the correlation of preoperative and intraoperative parameters with primary anatomical success. The secondary outcome was the correlation of various factors with age and final visual outcome.
Measured decimal VA was converted to logMAR for statistical analysis. Statistical analysis of univariate correlations was performed using Mann-Whitney U-tests, chisquare tests, or Fisher exact tests. Binary logistic regression models were used to identify independent risk factors for surgical failure. All statistical analyses were performed using IBM SPSS ver. 21.0 (IBM Corp., Armonk, NY, USA), setting the level of statistical significance at p < 0.05.
A total of 127 eyes from 127 patients were included. A summary of baseline characteristics is shown in Table 1. After the first operation, 108 eyes (85.0%) were successfully reattached. Nineteen eyes (15.0%) required a second operation due to failed primary surgery and were reattached without further operation in all cases. Vitrectomy and buckle revision were performed as the second operation in 12 and seven eyes, respectively. Five (3.9%) underwent vitrectomy due to macular complications during follow-up.
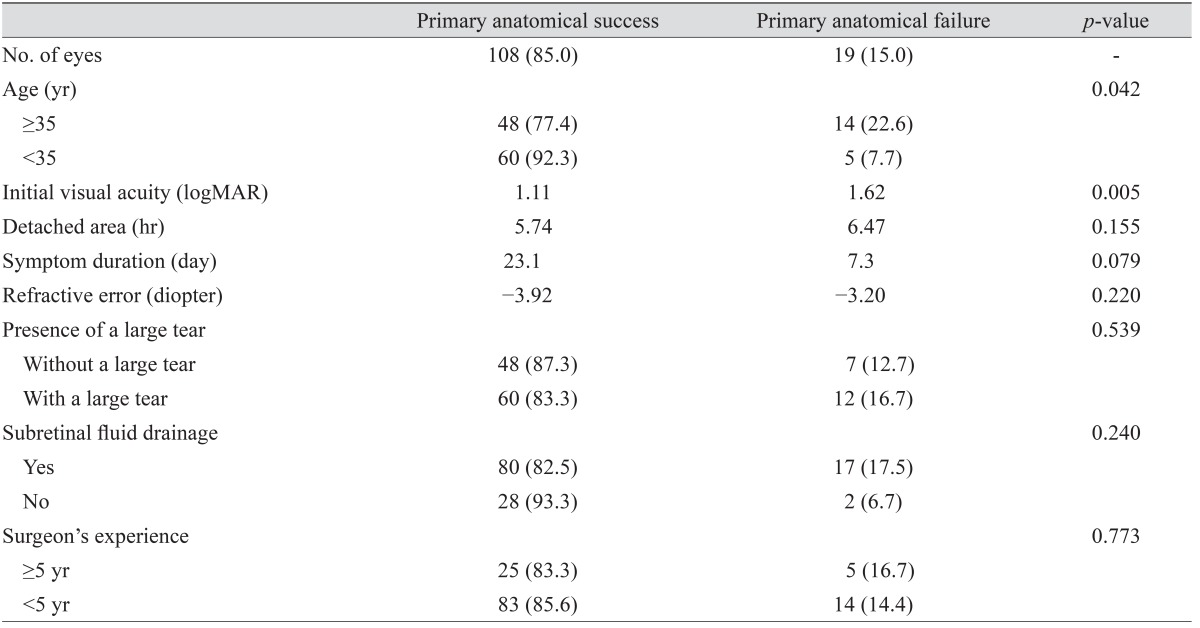
Primary anatomical failure occurred more frequently in older patients (p = 0.042) and those with lower VA (p = 0.005). There was no significant difference in the anatomical success rates with respect to detached area (p = 0.155), symptom duration (p = 0.079), refractive error (p = 0.220), presence of a large tear (p = 0.539), or SRF drainage (p = 0.240). These findings are summarized in Table 2.
In binary logistic regression analysis, except for preoperative VA, is not considered a single factor, older age (≥35) was the sole independent risk factor for primary anatomical failure (odds ratio, 3.5; 95% confidence interval, 1.201 to 10.940; p = 0.022).
The impact of age on SB was investigated by comparing various parameters between the older (≥35) and younger age (<35) groups (Table 3).
Mean preoperative VA (logMAR) was 1.44 in the older age group, which was significantly worse than 0.94 in the younger age group (p < 0.001). Higher postoperative visual improvement was observed in the older age group (p = 0.003), and there was no significant difference in final VA between the two groups. The primary anatomical success rate was 77.4% in the older age group, which was lower than the 92.3% in the younger age group (p = 0.042). The older age group had shorter symptom duration, less myopic eyes, more frequent large tears, and more frequent SRF drainage than the younger age group. Three cases of macular hole and two cases of epiretinal membrane occurred, but only in the older age group (p = 0.048). Sustained SRF was found at 3 months in 26.7% of the older age group versus 51.2% of the younger age group (p = 0.028).
No significant differences in detachment area and follow-up period were observed between age groups.
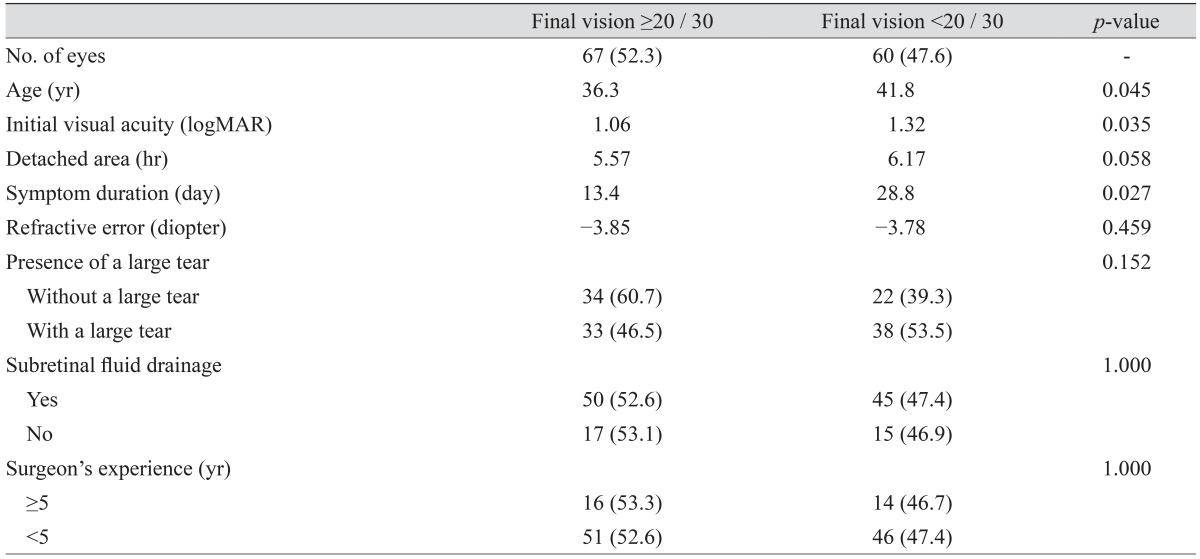
The eyes with better visual outcome (≥20 / 30) had better initial VA acuity (mean logMAR, 1.06; p = 0.035), were a younger age (mean, 36.3 years; p = 0.045), and had shorter symptom duration (mean, 13.4 days; p = 0.027) (Table 4). There was no significant difference in better visual outcome with respect to the detached area (p = 0.058), refractive error (p = 0.459), presence of a large tear (p = 0.152), or SRF drainage (p = 1.000) (Table 4).
In the current study, older age (≥35) was the only independent risk factor for anatomical failure after SB for uncomplicated phakic RRD in a binary logistic regression model, aside from preoperative VA. Although initial VA also showed a significant correlation with anatomical success on univariate analysis, it may be influenced by multiple factors, and was therefore not considered as a single factor. Accordingly, VA was excluded from the binary logistic regression analysis.
Several reports have implied that age is a prognostic factor in RRD repair. Wong et al. [7] reported that older age was a negative factor for final anatomical success and functional success in RRD. Cheng et al. [8], who investigated surgical outcomes for RRD in high myopic eyes, reported that young age was the only significant factor for anatomical success using multivariate analysis. However, the previous studies included heterogeneous patients who not only underwent SB, but also vitrectomy or combined surgery. Our study reaffirmed that age is a significant factor for anatomical outcomes in SB surgery.
Park et al. [9] reported that RRD in Korea is characterized by the bimodal distribution of the incidence according to age, with two peak incidence age groups (20-29 years and 60-69 years), and its inflection was around 35 years of age. A bimodal pattern was also found in the current study (Fig. 1) and a study conducted in Germany [10]. Our data showed a younger peak (20 to 29), which seemed to be associated with myopia rather than posterior vitreous detachment (PVD) in RRD. Mitry et al. [11] reported that RRD patients without PVD were younger and more myopic, supporting our results.
Several characteristics of RRD were evaluated to explain the correlation between age and anatomical outcomes. Poor initial vision, less myopic eyes, shorter symptom duration, and presence of a large tear were more common in the older age group. All of these correlations could be explained by vitreous liquefaction resulting in PVD. RRD in the young age group showed characteristics of less liquefied vitreous (Fig. 2), which tends to progress slowly and to have shallow detachment and was reported to show a correlation with better preoperative VA [12]. Less visual loss and slow progression could be related to late recognition and longer symptom duration. On the other hand, RRD in the older age group (Fig. 3) demonstrated features associated with PVD. For instance, they had poor initial vision, large tears, and faster progressing bullous RRD, suggesting strong vitreo-retinal traction. Despite poor preoperative VA, visual recovery was greater in the older age group than in the younger age group that had longer symptom duration, suggesting poor visual recovery [13]. There was no difference in postoperative VA between the two age groups.
Postoperative macular complications also support the above explanations. PVD-related complications, such as macular hole and epiretinal membrane [14], were only identified in the older age group. The prevalence of development of a macular hole after RRD repair was reported to be from 0.5% to 2.0%. Most of these patients (85% to 100%) were over 36 years old [15,16,17]. Previous studies have demonstrated that epiretinal membrane after RRD repair occurred more commonly in old age [18], whereas sustained SRF was more frequent in the young age group, which could be related to long symptom duration [19].
The lower primary reattachment rate in the older age group has a number of potential explanations. Non-liquefied vitreous could act as a mechanical barrier, blocking the passage of fluid through a break (Fig. 4A). However, PVD with liquefied vitreous (Fig. 4B) can counteract the buckle effect. In addition, a stronger traction related with PVD may be prone to a new break. These hypotheses imply that conserving the vitreous (via SB) would be a better method for less liquefied vitreous, while removing the vitreous (via PPV) would be better for liquefied vitreous.
Primary anatomical failure increased dramatically in patients over 35 years of age (Fig. 1). Additionally 35 years of age was the inflection point of bimodal RRD incidence in Korea [9]. Therefore, the study population was divided into two groups, above and below 35 years of age, to analyze the correlation with anatomical success. However, 35 years of age should not be considered as the definitive criterion for dividing patients by vitreous status or for surgical indication. Furthermore, myopia is more common and more severe in East Asia [20], and thus the current reference age (35 years of age) might be different in other ethnic groups.
The primary SB success rate may be influenced by the study design, which is related to case simplicity, lens status, or randomization. In retrospective studies, high success rates tend to be reported (as high as 88.8% to 92%) [7,21,22], while in large randomized prospective studies, success rates as low as 63.6% to 80% are reported [3,23]. Lower success rates, from 53.4% to 76%, have been reported for pseudophakic eyes [3,4]. The current study retrospectively examined phakic eyes and utilized a study design similar to that of Wong et al. [7] and Miki et al. [21]. Our study included the additional criterion of macular-off RRD, which showed a correlation with low success [24], and the overall success rate (85.9%) was lower than other similar studies [7,21].
The current study has several limitations, including its retrospective design, short follow-up period, and a small number of patients from a single center. The inclusion criterion of 3-month follow-up is too short to reflect the final functional outcome; however, it is likely sufficient to assess anatomical outcome and may reduce selective bias. Although it is expected that direct evaluation of vitreous liquefaction would provide more reliable prognostic information, there is no standard method for their accurate evaluation of PVD, with the exception of vitrectomy in fovea off RRD.
In summary, there was one feature that could differentiate between older and younger RRD: older age (≥35) was an independent prognostic factor for primary anatomical failure in SB for RRD. Vitreous liquefaction and posterior vitreous detachment are the likely underlying mechanisms. PPV would be a viable treatment option for older patients or those with characteristics of vitreous liquefaction/PVD. Further studies are required in order to compare the efficacy of SB and PPV for in such RRD.
Acknowledgements
This work was supported by clinical research grant from Pusan National University Hospital in 2015.
The authors thank to Han Yong Bae and Hyun Jin Park for acquiring and collecting the optical coherent tomography data.
Notes
Conflict of Interest: No potential conflict of interest relevant to this article was reported.
REFERENCES
1. Pastor JC, Fernandez I, Rodriguez de la Rua E, et al. Surgical outcomes for primary rhegmatogenous retinal detachments in phakic and pseudophakic patients: the Retina 1 Project. Report 2. Br J Ophthalmol 2008;92:378-382.


2. Afrashi F, Akkin C, Egrilmez S, et al. Anatomic outcome of scleral buckling surgery in primary rhegmatogenous retinal detachment. Int Ophthalmol 2005;26:77-81.


3. Heimann H, Bartz-Schmidt KU, Bornfeld N, et al. Scleral buckling versus primary vitrectomy in rhegmatogenous retinal detachment: a prospective randomized multicenter clinical study. Ophthalmology 2007;114:2142-2154.


4. Sharma YR, Karunanithi S, Azad RV, et al. Functional and anatomic outcome of scleral buckling versus primary vitrectomy in pseudophakic retinal detachment. Acta Ophthalmol Scand 2005;83:293-297.


5. Feltgen N, Heimann H, Hoerauf H, et al. Scleral buckling versus primary vitrectomy in rhegmatogenous retinal detachment study (SPR study): risk assessment of anatomical outcome. SPR study report no. 7. Acta Ophthalmol 2013;91:282-287.


6. Schneider EW, Geraets RL, Johnson MW. Pars plana vitrectomy without adjuvant procedures for repair of primary rhegmatogenous retinal detachment. Retina 2012;32:213-219.


7. Wong CW, Wong WL, Yeo IY, et al. Trends and factors related to outcomes for primary rhegmatogenous retinal detachment surgery in a large Asian tertiary eye center. Retina 2014;34:684-692.


8. Cheng SF, Yang CH, Lee CH, et al. Anatomical and functional outcome of surgery of primary rhegmatogenous retinal detachment in high myopic eyes. Eye (Lond) 2008;22:70-76.


9. Park SJ, Choi NK, Park KH, Woo SJ. Five year nationwide incidence of rhegmatogenous retinal detachment requiring surgery in Korea. PLoS One 2013;8:e80174



10. Thelen U, Amler S, Osada N, Gerding H. Success rates of retinal buckling surgery: relationship to refractive error and lens status: results from a large German case series. Ophthalmology 2010;117:785-790.


11. Mitry D, Singh J, Yorston D, et al. The predisposing pathology and clinical characteristics in the Scottish retinal detachment study. Ophthalmology 2011;118:1429-1434.


12. Hagimura N, Suto K, Iida T, Kishi S. Optical coherence tomography of the neurosensory retina in rhegmatogenous retinal detachment. Am J Ophthalmol 2000;129:186-190.


13. Burton TC. Recovery of visual acuity after retinal detachment involving the macula. Trans Am Ophthalmol Soc 1982;80:475-497.


14. Shechtman DL, Dunbar MT. The expanding spectrum of vitreomacular traction. Optometry 2009;80:681-687.


15. Moshfeghi AA, Salam GA, Deramo VA, et al. Management of macular holes that develop after retinal detachment repair. Am J Ophthalmol 2003;136:895-899.


16. Benzerroug M, Genevois O, Siahmed K, et al. Results of surgery on macular holes that develop after rhegmatogenous retinal detachment. Br J Ophthalmol 2008;92:217-219.


17. Garcia-Arumi J, Boixadera A, Martinez-Castillo V, et al. Macular holes after rhegmatogenous retinal detachment repair: surgical management and functional outcome. Retina 2011;31:1777-1782.


18. Uemura A, Ideta H, Nagasaki H, et al. Macular pucker after retinal detachment surgery. Ophthalmic Surg 1992;23:116-119.


19. Veckeneer M, Derycke L, Lindstedt EW, et al. Persistent subretinal fluid after surgery for rhegmatogenous retinal detachment: hypothesis and review. Graefes Arch Clin Exp Ophthalmol 2012;250:795-802.


21. Miki D, Hida T, Hotta K, et al. Comparison of scleral buckling and vitrectomy for retinal detachment resulting from flap tears in superior quadrants. Jpn J Ophthalmol 2001;45:187-191.


22. Tani P, Robertson DM, Langworthy A. Prognosis for central vision and anatomic reattachment in rhegmatogenous retinal detachment with macula detached. Am J Ophthalmol 1981;92:611-620.


Fig. 1
Age of patients with rhegmatogenous retinal detachment. Data shows bimodal distribution with double peaks. Anatomical failure after primary scleral buckle surgery was observed more frequently in patients older than 35 years (p = 0.042).
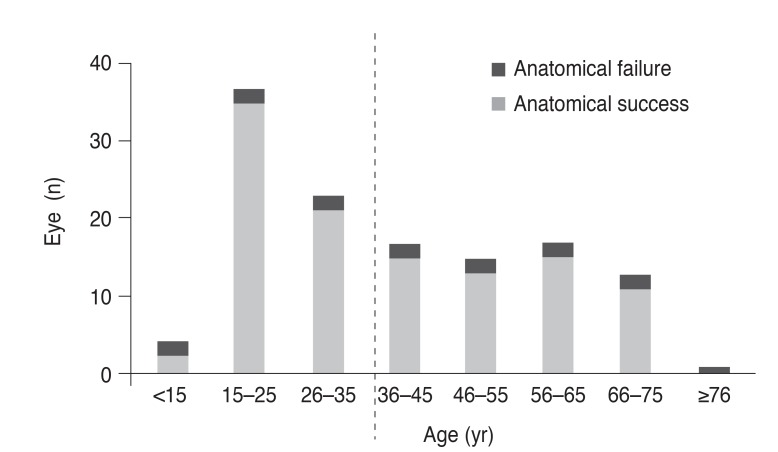
Fig. 2
Case 1: typical fundus findings of a young patient with rhegmatogenous retinal detachment (less liquefied without posterior vitreous detachment, long symptom duration, better initial visual acuity, small retinal tear, lower visual recovery, and sustained subretinal fluid). A 20-year-old male visited Pusan National University Hospital complaining of decreased visual acuity of his left eye for more than 1 month. His best-corrective visual acuity was 20 / 200 in that eye. Fundus photo (left) shows shallow retinal detachment (white dotted line) with a small retinal hole (black line) and a subretinal strand. The retina was reattached after scleral buckling without any adjuvant procedure (upper right). His best-corrected visual acuity improved to 20 / 50 and optical coherent tomography (lower right) shows sustained subretinal fluid at 3 months after surgery.
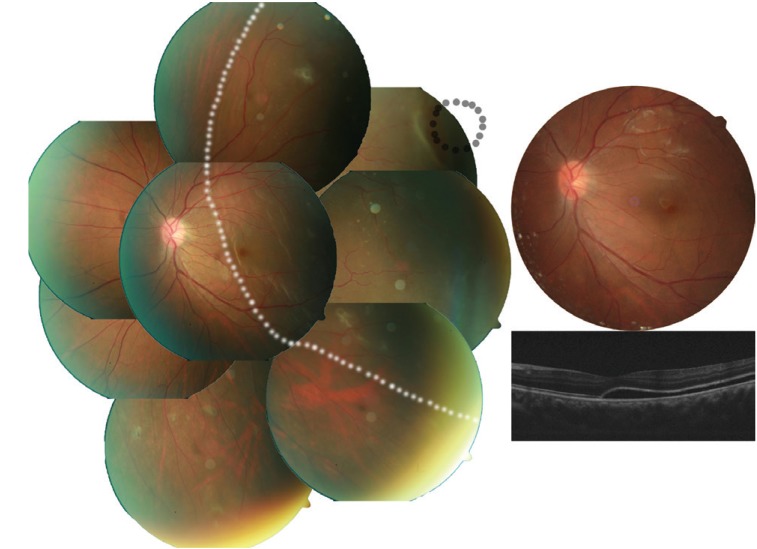
Fig. 3
Case 2: typical fundus finding of an older patient with rhegmatogenous retinal detachment specified by liquefied with posterior vitreous detachment, short symptom duration, poor initial visual acuity, large retinal tear, better visual recovery, and needed subretinal fluid (SRF) drainage. A 60-year-old male visited Pusan National University Hospital complaining of sudden visual loss of his left eye 4 days ago. His best-corrected visual acuity was measured by counting finger in counting for that eye. Fundus photo (left) shows bullous retinal detachment (white dotted line) with a large tear of 1.0 disc diameter (black line). The retinal tear could not be settled on the retinal pigment epithelium by scleral protrusion. After SRF draining, the tear faced the pigment epithelium due to the buckle effect. His best-corrected visual acuity improved to 10 / 20 and there was no SRF on fundus photographs (upper right) or optical coherent tomography (lower right) 2 months after surgery.
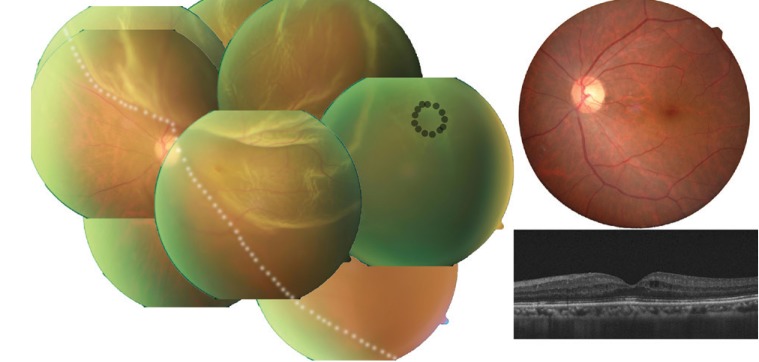
Fig. 4
Theory of vitreous roles in rhegmatogenous retinal detachment. Less-liquefied vitreous (A) could play a role as a mechanical barrier like tamponade that blocks the passage of fluid through a break. However, liquefied vitreous with posterior vitreous detachment (B) can counteract the buckle effect.
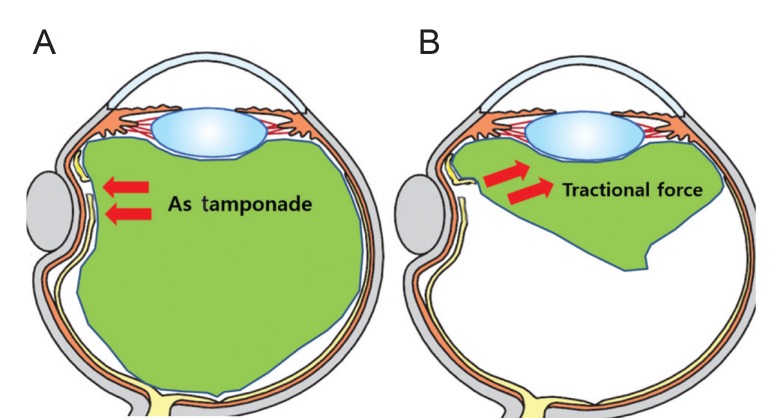
Table 2
Results of univariate analysis of various parameters for anatomical success after scleral buckle in rhegmatogenous retinal detachment
- TOOLS
-
METRICS

-
- 12 Crossref
- 0 Scopus
- 3,252 View
- 17 Download
- Related articles
-
Neovascular Glaucoma Associated with Chronic Rhegmatogenous Retinal Detachment2023 June;37(3)
Reattachment of Rhegmatogenous Retinal Detachment via Fibrin Tissue Adhesive2021 June;35(3)
Changing Trends in Surgery for Retinal Detachment in Korea2014 December;28(6)
Macular Hole Formation in Rhegmatogenous Retinal Detachment after Scleral Buckling2014 October;28(5)




 PDF Links
PDF Links PubReader
PubReader Full text via DOI
Full text via DOI Full text via PMC
Full text via PMC Download Citation
Download Citation Print
Print



