Intraocular silicone oil (SO) to treat complicated retinal detachment (RD) was first introduced in 1962 [
1]. In the early 1970s, several investigators used SO in cases with proliferative retinopathy (PVR) [
2,
3,
4,
5,
6,
7]; since then, it has been applied frequently as an internal tamponade for complex cases. This approach can also be used for surgical management of proliferative diabetic retinopathy (PDR); however, some authors have reported that SO has questionable benefits [
8], while others have advocated its use to promote retinal stabilization and iris neovascularization regression [
9,
10]. It has also been reported to be helpful for maintaining postoperative clear media [
8] and more rapid visual rehabilitation [
9,
11]. It has been demonstrated that the anatomical success of vitrectomy and SO injection in cases with PDR is similar to cases with PVR (70% and 67%, respectively); however, visual results were quite different. Compared to eyes with PVR, a significantly larger group of patients with PDR failed to recover final visual acuity of 5 / 200 or better and did not achieve light perception [
8]. Additionally, SO is associated with significant complications such as cataract, band keratopathy, corneal decompensation, ocular hypotony, and recurrent RD resulting from reproliferation. Therefore, the role of SO in the management of severe PDR is less clear than its role in PVR.
In this study, we evaluated the visual and anatomical outcomes of vitrectomy plus SO injection in eyes with advanced PDR. We also assessed factors that affect outcomes. This retrospective study provided information about indications for SO injections, type of SO used, time of its removal, and the rate of complications in our centers.
Materials and Methods
This was a retrospective, two-center study and included all eyes that underwent vitrectomy plus SO injection to manage advanced PDR in two university-affiliated hospitals (Labbafinejad and Imam Hossein Medical Centers) between 2004 and 2013, with more than 3 months of follow-up. This study was approved by the review board/ethics committee of the Ophthalmic Research Center of Shahid Beheshti University of Medical Sciences.
Operations were performed under local or general anesthesia using a standard 3-port 20-gauge pars plana vitrectomy technique through sclerotomy 3.5 to 4 mm posterior to the limbus. Phacoemulsification with or without intraocular lens (IOL) implantation was performed during the same session, when indicated. The pupil was dilated with iris retractors when necessary. Other complementary vitreoretinal procedures such as segmentation/delamination, fibrovascular tissue dissection, perfluorocarbon liquid injection, endocauterization, and endolaser photocoagulation were performed as needed. In all cases, SO was injected either manually or by pump through one of the sclerotomy sites following fluid/air exchange. Three types of SO were used according to the surgeons' discretion: 1,000 centistokes (cs), 5,000 to 5,700 cs, and heavy (high density) SO. Sclerotomy sites and conjunctiva openings were sutured with Vicryl 7-0. At the end of the procedure, all eyes received a subconjunctival injection of 20 mg betamethasone and 100 mg cefazolin. Postoperative medications included topical antibiotics for 5 days, in addition to cycloplegic and corticosteroid drops that were tapered for a period of 4 to 6 weeks postoperatively.
Preoperative and postoperative data from patients' files including best-corrected visual acuity (BCVA), presence of relative afferent pupillary defect, slit-lamp examination findings, intraocular pressure (IOP) measured by Goldman applanation tonometry, and fundus findings were recorded for analysis. Lens opacity was graded from 0 to 4+ in three main categories of nuclear sclerosis, posterior subcapsular, and cortical cataract, according to the Lens Opacities Classification System II [
12]. Baseline macular status, i.e., attached or detached, was determined by ophthalmoscopy and echographic findings if needed. Indications for vitrectomy, reasons for SO injection, type of SO used, and the time of SO removal were also recorded.
In this study, visual and anatomical outcomes, the rate and time of SO removal, and potential complications after surgical intervention were evaluated. Postoperative retinal status was categorized as complete attachment, partial attachment (only less than one fourth of the retina outside of the macula remained detached), and no attachment. To evaluate the effect of various factors on anatomical outcomes, however, partial attachment was also considered anatomical failure.
The Snellen chart for BCVA measurement was used and converted to the logarithm of the minimum angle of resolution (logMAR) for statistical evaluation. Additionally, BCVA changes were evaluated by reporting the percentage of eyes with BCVA Ōēź20 / 200 or <20 / 200 Snellen visual acuity before the operation and at the final visit. The BCVA changes were also classified into worsened, stable, and improved based on at least two Snellen line changes compared to the baseline value.
Statistical analysis
To describe data, the mean, standard deviation, median and range, and frequency and percentage were calculated as appropriate. The Mann-Whitney, chi-square, and Fisher's exact tests were used to compare the results between the groups. The Wilcoxon singed-rank test was used to evaluate the changes within the groups. The generalized estimating equation was used to compare the groups adjusted for baseline or simultaneous effects of different variables when considering the possible correlation of two eyes in a subject. All statistical analyses were performed by SPSS ver. 22.0. (IBM Corp., Armonk, NY, USA). A p-value less than 0.05 was considered statistically significant.
Results
A total of 236 eyes of 213 patients (40.4% male and 59.6% female) with a mean age of 52.6 ┬▒ 11.1 years (range, 21 to 79) who underwent vitrectomy plus SO injection for complications of PDR were included. Of all the patients, 188 (88.3%) had non-insulin-dependent diabetes mellitus, and 25 (11.7%) had insulin-dependent diabetes mellitus. Seventy-five patients (35.2%) were hypertensive and were receiving appropriate treatment. Six cases (2.8%) suffered from renal failure and four had regular hemodialysis. One patient had a history of renal transplantation. The mean follow-up time was 88.4 ┬▒ 58.1 weeks (median, 74; range, 12 to 341).
Before vitrectomy plus SO injection, 196 eyes (83.1%) had undergone panretinal laser therapy; four eyes (1.7%) had a history of vitrectomy; and 23 eyes (9.7%) had undergone cataract surgery (
Table 1). Relative afferent pupillary defect was positive in 39 cases (16.5%) preoperatively, and macular detachment was detected in 127 eyes (53.8%). According to the preoperative files, the indications for vitrectomy were: (1) non-clearing dense vitreous hemorrhage (VH) in four eyes (1.7%), (2) tractional retinal detachment (TRD) involving or threatening the macula in 83 eyes (35.2%), (3) TRD based on echographic findings associated with non-clearing dense VH in 37 eyes (15.7%), (4) combined rhegmatogenous and tractional RD (combined RD) in 52 eyes (22.0%), and (5) severe progressive fibrovascular proliferation with or without TRD in 60 eyes (25.4%).
Combined vitrectomy and cataract surgery was performed in 18 eyes (7.6%) (
Table 1). Of these, 16 eyes (88.8%) underwent IOL implantation. The main cataract surgery method was phacoemulsification, and extracapsular cataract extraction was performed in only one eye (0.4%).
Three types of SO were used, according to the surgeons' preference: 1,000 cs SO in 80 eyes (33.9%), 5,000 (or 5,700) cs SO in 148 eyes (62.7%), and heavy (high density) SO in eight eyes (3.4%). There was one or multiple reasons for SO injection in each eye. The main reasons for SO injection according to the intraoperative findings were: (1) curved retina after removal of all tractions in 142 eyes (60.2%), (2) combined RD in 37 eyes (15.7%), and (3) intraoperative iatrogenic retinal breaks in 57 eyes (24.1%). Iatrogenic breaks were most frequently located posteriorly (56 eyes, 98.2%) rather than on the anterior retina, which occurred in one eye (1.8%) posterior to the sclerotomy site.
The studied eyes were divided into two groups. Group 1 consisted of 155 eyes (65.7%) that underwent SO removal before the final visit. The mean time for SO removal in this group was 53.0 ┬▒ 30.9 weeks (median, 49; range, 5 to 172). The mean follow-up after SO removal was 42.9 ┬▒ 53.6 weeks (median, 21; range, 1 to 308), and the mean total follow-up time for this group was 96.2 ┬▒ 56.0 weeks (median, 82; range, 23 to 314). At the final visit before SO removal, 95 eyes (61.3%) had signs of SO emulsification, and 15 eyes (9.7%) demonstrated increase in IOP. In this group, 80 eyes (51.6%) were candidates for accompanying cataract surgery. Cataract surgery was performed with or without IOL implantation in 72 and eight eyes (46.5% and 5.1%), respectively. Moreover, two eyes (1.3%) underwent epimacular membrane removal during the SO removal session.
Group 2 consisted of 81 eyes (34.3%) with intact SO at the final visit. In 79 eyes, SO removal was not performed because of failure to achieve a stable retina. The other two eyes underwent another vitreoretinal surgery with reinjection of SO. The mean follow-up for this group was 71.7 ┬▒ 59.3 weeks (median, 52; range, 12 to 341). Cataract surgery without SO removal was performed with and without lens implantation in one (1.3%) and two (2.5%) eyes, respectively, in this group.
Anatomical results
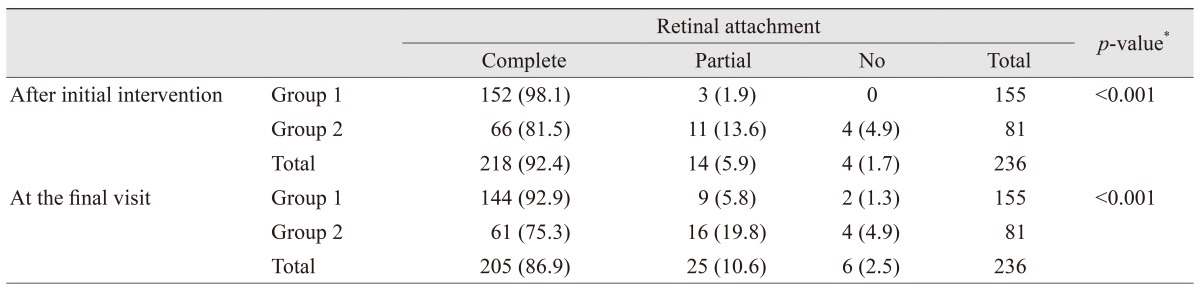
After initial intervention (pars plana vitrectomy with SO injection), complete, partial, and no retinal attachment were observed in 218 (92.4%), 14 (5.9%), and four (1.7%) eyes, respectively. The features of each group are presented in
Table 2. In group 1, after SO removal (with or without other procedures), nine eyes (5.8%) had partial RD, and two eyes (1.3%) developed total RD. For all the studied eyes and at the final visit, complete, partial, and no retinal attachment were observed in 205 (86.9%), 25 (10.6%), and six (2.5%) eyes, respectively. The difference in anatomical outcomes between the groups was significant both after initial intervention and at the final visit (
p < 0.001 for both), with superior outcomes noted in group 1.
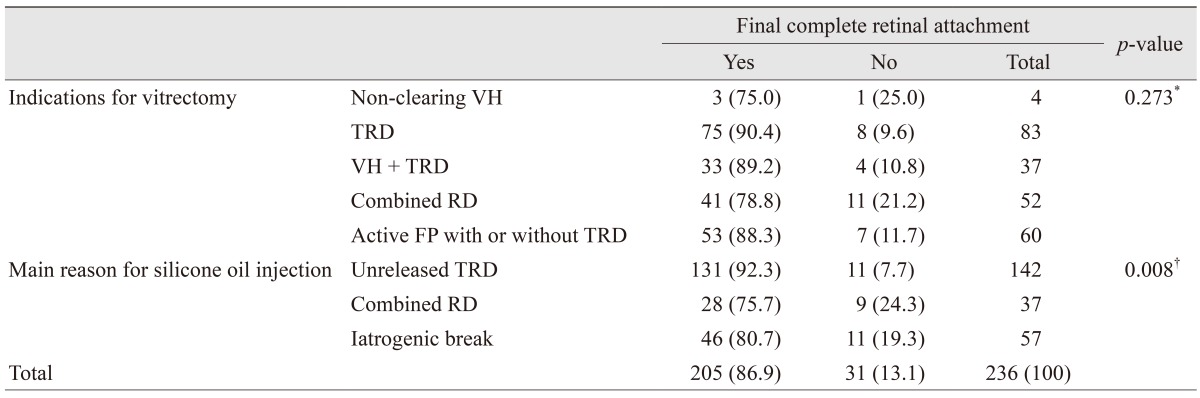
Table 3 shows the relationships between the indications for vitrectomy and reasons for SO injection with final anatomical outcomes. Although eyes with preoperative indications of non-clearing VH or combined RD had a lower risk of final complete retinal attachment (75.0% and 78.8%, respectively), the difference in indicators in terms of final complete attachment was not statistically significant (
p = 0.273). Nonetheless, the three main intraoperative reasons for SO injections had significantly different effects on the final anatomical outcome (
p = 0.008). Pairwise comparison showed that the two main reasons for combined RD and development of iatrogenic breaks were significantly (
p = 0.025 and
p = 0.042, respectively) associated with a decreased risk of final complete retinal attachment (75.7% and 80.7%, respectively) compared to unreleased TRD (92.3%).
Cases with heavy SO were significantly associated with a higher rate of anatomical failure at the final visit (
p = 0.016). Complete final retinal attachment was observed in 86.3% and 89.2% of the eyes filled with 1,000 and 5,000 cs SO at the initial surgery, respectively, while this occurred in only 50% of the eyes with heavy SO (
Table 4). Considering each group separately, the difference in SO type was not associated with final anatomical success (
p = 0.227) in group 1 but was indicated in group 2 (
p = 0.022). In group 1 (
Table 5), SO removal was performed within 6 months of injection in 32 eyes, 20.6%; between 6 and 12 months in 58 eyes, 37.4%; and more than 12 months after injection in 65 eyes, 42.0%. The anatomic outcome in this group was not statistically significantly with regard to time of SO removal (
p = 0.848).
Visual results
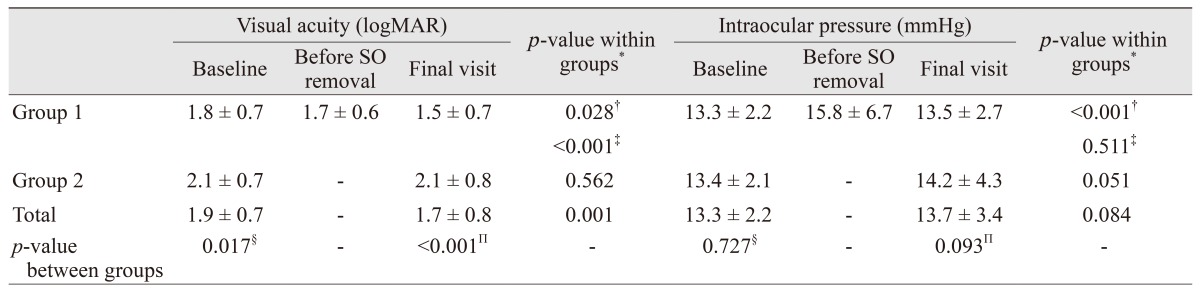
The mean BCVA was 1.9 ┬▒ 0.7 logMAR at the initial presentation and significantly improved to 1.7 ┬▒ 0.8 logMAR at the final follow-up visit (
p = 0.001) (
Table 6). The BCVA improvement was significant in group 1 both before SO removal and at the final visit (
p = 0.028 and
p < 0.001, respectively). In group 2, however, the final mean BCVA was not statistically significantly different from the baseline value (
p = 0.562). The initial mean BCVA in group 2 was significantly lower than that of group 1 (
p = 0.017).
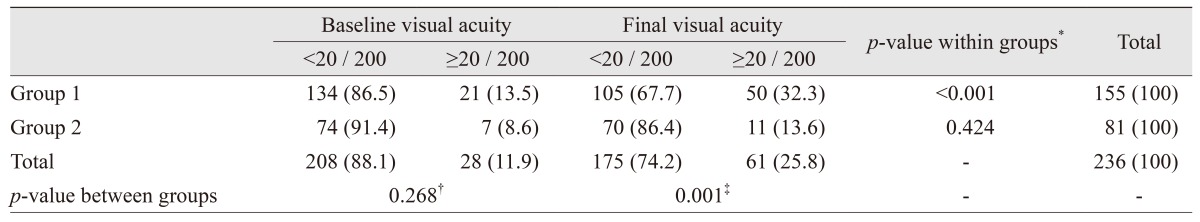
At presentation, the number of eyes with BCVA Ōēź20 / 200 was 28 (11.9%) and increased to 61 (25.8%) at the final visit (
p = 0.002) (
Table 7). The difference in the percentage of eyes with BCVA Ōēź20 / 200 at baseline between the groups was not significant (
p = 0.268). However, more eyes in group 1 (32.3%), achieved BCVA Ōēź20 / 200 than the eyes in group 2 (13.6%). This difference between the groups was statistically significant (
p = 0.002).
In a different type of functional outcome evaluation, BCVA change in each eye was classified to improved, remain stable, or worsen based on an at least two Snellen line change (
Table 8). The analysis indicated that 51.7% of eyes experienced at least two lines of improvement, while 27.5% showed the same level or worsening. Comparing the two groups regarding this type of classification, the difference was statistically significant (
p = 0.002), with group 1 demonstrating better outcomes.
Multivariate analyses
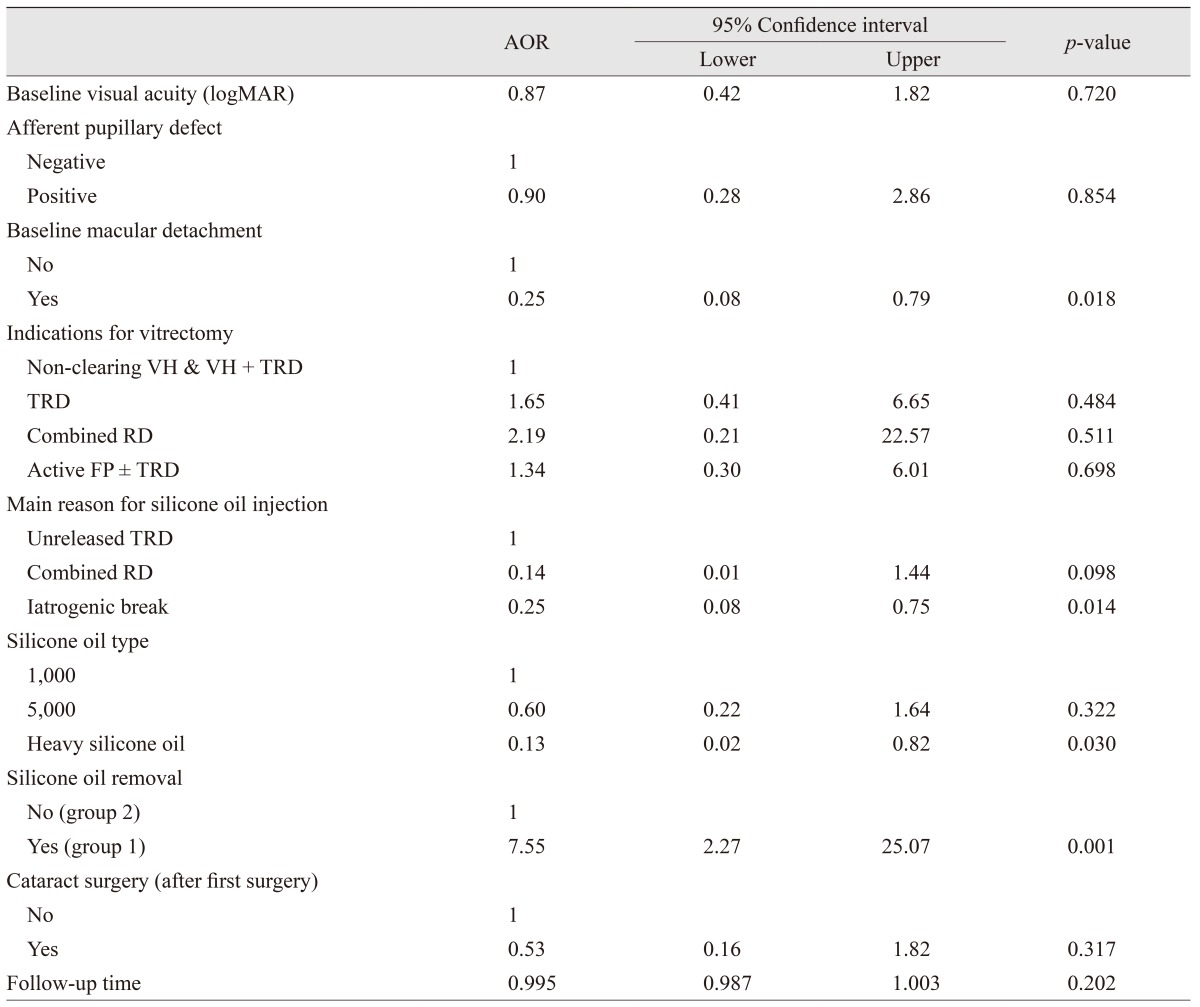
Multivariate analysis was performed to identify possible factors for predicting anatomical and visual outcomes. Since the number of cases with initial diagnosis of pure VH was limited (four eyes), these cases were combined with the VH + TRD cases in the regression models. Considering the final complete attachment of the retina, macular detachment at baseline (adjusted odds ratio [AOR], 0.25;
p = 0.018), development of iatrogenic break (AOR, 0.25;
p = 0.014), and use of heavy SO (AOR, 0.13;
p = 0.030) were independently associated with lower risk of final complete retinal attachment, and SO removal (group 1) was correlated with a higher risk of retinal attachment (AOR, 7.55;
p = 0.001) (
Table 9).
Functional outcome analysis indicated that BCVA improvement from baseline to the final visit was positively associated with lower baseline BCVA and SO removal and negatively with presence of macular detachment at baseline (
Table 10). Considering final BCVA Ōēź20 / 200 as the outcome in multivariate analysis, presenting BCVA was the only independent factor (
p = 0.001); i.e., eyes with better initial BCVA had a greater chance of BCVA Ōēź20 / 200 at the final visit (data not shown).
Complications
The mean IOP was 13.3 ┬▒ 2.2 mmHg at baseline and 13.7 ┬▒ 3.4 mmHg at the final visit (
p = 0.084) (
Table 6). In group 1, mean IOP increased significantly from 13.3 ┬▒ 2.2 mmHg at the initial visit to 15.8 ┬▒ 6.7 mmHg before SO removal (
p < 0.001). However, it decreased to 13.5 ┬▒ 2.7 mmHg (
p = 0.511, compared to the baseline) at the final follow-up. There were no statistically significant differences in the initial and final values of mean IOP between the groups (
p = 0.727 and
p = 0.093, respectively).
After initial operations, 34 eyes (14.4%) had an IOP >21 mmHg and received medical treatment. In four eyes, IOP was not controlled with medication, and early SO removal was performed. In one case, IOP was not normalized even after SO removal, and antiglaucoma medications were continued. In 19 eyes with IOP >21 mmHg, SO removal was performed during the follow-up; in nine cases, IOP returned to a normal value without any medication. In total, 21 eyes (8.9%) needed antiglaucoma medications at the final visit, 10 eyes in group 1 and 11 in group 2.
The progression of lens opacity in all categories was statistically significant up to the time of SO removal in group 1 and up to the time of the final visit in group 2. The mean grades of lens opacity in the three categories of nuclear sclerosis, posterior subcapsular, and cortical cataract for group 1 were 1.6 ┬▒ 0.5, 0.4 ┬▒ 0.6, and 0.4 ┬▒ 0.6, respectively, at baseline and progressed to 2.4 ┬▒ 0.8 (p < 0.001), 1.3 ┬▒ 1.1 (p < 0.001), and 0.6 ┬▒ 0.8 (p = 0.001) before SO removal. These values for group 2 were 1.7 ┬▒ 0.6, 0.4 ┬▒ 0.6, and 0.3 ┬▒ 0.7, respectively, at baseline and progressed to 2.3 ┬▒ 0.7 (p < 0.001), 0.9 ┬▒ 1.1 (p < 0.001), and 0.6 ┬▒ 0.8 (p = 0.004) at the final visit. Eighty-three eyes (42.5% of the remaining phakic eyes after primary intervention) underwent cataract surgery alone or surgery combined with other procedures during the follow-up time. Of these, 73 eyes underwent IOL implantation.
Table 11 presents the rates of complications (with the exception of ocular hypertension and lens opacity progression) up to the final visit in all cases and in each group. The most common adverse event was optic atrophy, which was noted in 26 eyes (16.8%) in group 1 and 14 eyes (17.3%) in group 2. The difference between the groups regarding the frequency of optic atrophy was not statistically significant (
p = 0.888).
Discussion
In this retrospective study, an anatomical success rate of 86.9% was identified in eyes that underwent vitrectomy plus SO injection for advanced PDR. An improvement in mean BCVA was also detected; however, this occurred mostly in eyes that underwent SO removal before the final visit. A visual result of Ōēź20 / 200 was independently associated only with initial BCVA, while final anatomical failure was correlated with macular detachment at baseline, development of iatrogenic break during surgery, and use of heavy SO. Increase in IOP and lens opacity progression were among the most common complications observed in these eyes and were managed successfully in most cases; optic atrophy also occurred in many cases.
Retinal redetachment or nonattachment after primary vitrectomy in PDR is usually severe and extensive and can render the affected eye inoperable [
13]. Use of SO with its long-acting tamponade effect has been shown to increase the retinal reattachment rate and anatomical success in cases with advanced PDR [
11,
14,
15]. In this study, final total retinal attachment was achieved in 86.9% of the eyes. This rate has been reported variously by different authors, ranging from 57% to 87% [
16]. This diversity can be explained by many factors; however, the most important is the difference in severity of preoperative retinopathy among different studies. Univariate analysis in our study showed that anatomical success decreased in the eyes with combined RD or in cases with iatrogenic retinal breaks. Therefore, either preoperative existence of retinal breaks or intraoperative formation could affect the result. Although an iatrogenic retinal break per se might worsen the outcome by adding a proliferative component, it can also indicate the severity of preoperative retinopathy. This factor is thus an important consideration for the operating surgeon. The rate of iatrogenic retinal breaks in this study was 24.1% and was comparable to some other studies, presenting rates of 20% [
17] and 28.5% [
18], and less than those ranging from 33% to 41% [
19,
20,
21]. Of note, the eyes reported in this article underwent 20-gauge vitrectomy. A shift to improved surgical techniques that employ newer vitrectomy machines with enhanced design of vitrectomy probes and high cut rates might decrease the risk of iatrogenic retinal break formation and hence have a positive effect on the anatomical outcome [
22].
In a retrospective study, Pavlovic et al. [
23] evaluated the long-term outcome after SO removal in 324 cases, including 87 eyes with PDR. The authors reported that the duration of SO tamponade had no effect on the redetachment rate. It was also noted that the time of SO removal had no effect on the final anatomical outcome. Earlier studies had shown no difference in tamponading force among purified SOs of various viscosities [
24,
25]. However, in a retrospective study on eyes with mixed etiologies, including PDR, Soheilian et al. [
26] concluded that, in complicated RD surgery, use of 5,000 cs SO might be associated with poorer anatomic and visual outcomes compared with use of 1,000 cs SO. The possible explanations for this observation are that the use of 5,000 cs oil in more complex cases and the possibility that eyes that had been filled with 5,000 cs oil were seen later in the disease process due to the longer retention period of the oil, thereby increasing the detection rate of retinal redetachment. It should be noted that most of the redetachments in Soheilian's study occurred to the PVR group. Such an explanation might be applicable to the observation of more anatomical failure in eyes with heavy SO in this study. This failure might be due to the greater initial complexity of the retinal disease, especially in the inferior retina, which prompted the surgeon to use heavy SO.
In similar studies, visual outcomes have been reported in different ways. In a retrospective study of 106 eyes with PDR treated by vitrectomy and SO injection, the following functional results were reported: satisfactory BCVA (0.5 to 0.05) in 23 eyes (22%), ambulatory vision (0.03 to counting fingers) in 14 eyes (13%), hand movement or light perception in 54 eyes (51%), and no light perception in 15 eyes (14%). In studies by Kim et al. [
27] and Castellarin et al. [
16], postoperative BCVA improved in 60% and 44% of patients, was unchanged in 20% and 12%, and worsened in 20% and 44%, respectively. The findings of the present study are comparable to these previous results; i.e., BCVA improvement (at least two Snellen lines) at the final visit compared to preoperative BCVA was noted in 51.7% of the eyes, while unchanged and worsened BCVA were observed in 20.8% and 27.5%, respectively. In cases with PDR, Karel and Kalvodova [
15] reported a BCVA Ōēź1 / 100 in 32% of operated eyes. Lucke et al. [
9] reported BCVA Ōēź5 / 200 in 69% of their complicated RD cases that underwent operation with SO; however, only 21% of these cases had PDR. In our study, a final BCVA Ōēź20 / 200 was achieved in 25.8% of the eyes.
Generally, final BCVA primarily depends on many baseline characteristics, in addition to some intraoperative and postoperative factors. Different authors have reported factors that can influence the functional results in different ways. Shen and Yang [
13] identified preoperative corrected BCVA as the only variable that was statistically significantly associated with final VA (
p < 0.001). In another report, functional failure was found to be caused by retinal redetachment in 47 eyes (43%), secondary glaucoma in 10 eyes (9%), retinal ischemia in 15 eyes (13%) and keratopathy in three eyes (3%) [
15]. It has also been reported that the functional results in these patients are limited by recurrent detachments due to characteristic reproliferations under the silicone bubble or by ischemic diabetic angiopathy in the attached retina [
28]. Multivariate analysis in this mentioned study showed that, in an eye with complex diabetic retinopathy, which was a candidate for vitrectomy, the only predictors for achieving final BCVA Ōēź20 / 200 was baseline BCVA. Based on our results, however, the two additional baseline parameters of lower initial BCVA and preoperative attached macula predicted visual improvement. It should be clarified that eyes with lower initial BCVA showed more improvement in BCVA, whereas eyes with better initial BCVA were more likely to achieve a final BCVA Ōēź20 / 200. The association of weaker initial vision with higher BCVA improvement might be explained by the statistical fact that eyes with lower baseline BCVA had more space for a greater change. However, macular ischemia might also be a strong factor that influences BCVA outcome but was not addressed in the current study.
In the present study, eyes that underwent SO removal were compared with that that had intact SO at the final visit. Both anatomical and functional outcomes were better in cases with removed SO. Therefore, SO retention in an eye with complex PDR might worsen the prognosis. Nonetheless, this difference might reflect the complexity of preoperative and/or postoperative PDR conditions in these eyes, which prompted the surgeons to maintain the SO in the eye. Additionally, neither early nor late SO removal affected final retinal attachment state. The same conclusion was presented in a study by Shen and Yang [
13] reporting the outcome of extended SO tamponade for complex PDR. They did not perform SO removal in any of their 45 cases and followed them for an average of 28.2 ┬▒ 15 months with the range of 12 to 69 months. Despite a general concern about the potential retinal toxicity from extended contact of SO with the retina, Shen and Yang showed no significant correlation between long-term BCVA change and follow-up duration.
There were two major complications of vitrectomy plus SO injection in our study: cataract formation and ocular hypertension. Since both were treatable in most of the cases, these complications do not need to be considered important factors in decision making. Cataract formation not only decreases vision, but also precludes a good posterior segment examination. It has been shown that early removal of SO during the first weeks after surgery in order to avoid cataract progression increases the likelihood of redetachment [
29]. Therefore, it is recommended that SO be kept in place even in cases of early cataract surgery [
29]. The previously reported rates of cataract formation in PDR cases with SO were 27% [
14] and 68% [
30] after about 12 months and 87.5% after 32 months [
31]. Significant cataract progression was observed in all of our cases during the follow-up period, and 42.5% of all phakic eyes after initial intervention underwent cataract surgery prior to the final visit.
Ocular hypertension, another complication that can result from use of SO in PDR cases, could be due to SO emulsification [
32]. The mean IOP increase (about 2 mmHg) in our cases was statistically significant. In total, 14.4% of cases needed antiglaucoma medications, and uncontrolled ocular hypertension necessitated early SO removal in four eyes. In about half of our cases with postoperative ocular hypertension, IOP did not return to the normal level after SO removal. The main reason for this refractory ocular hypertension is not well known but could be due to iris neovascularization [
32]. This occurrence was noted in 0.8% of our cases and 6% of cases in the study by Sima and Zoran [
14]. Some authors believe that SO can prevent iris neovascularization by improving oxygen transfer to the anterior segment of the eye [
10]. This beneficial effect, however, was not reported in another study [
8]. In addition, two of our cases developed iris neovascularization despite having intact SO.
Another relatively common sign noted in our patients was optic atrophy. Its prevalence was reported to be 16% [
11] and 10% [
9] in other studies. In diabetic eyes, optic and retinal atrophy could be the major causes of poor visual function and progressive loss of vision despite an attached retina [
9]. It has been hypothesized that SO in the eye can cause optic atrophy [
33]; nevertheless, optic atrophy can be a consequence of diabetic retinopathy [
34], diabetic papillopathy [
35], coexisting hypertension [
36], and vitrectomy [
37].
Ocular hypotony after complex RD surgery has been reported in 21% of cases by Azen et al. [
30]. Nonetheless, this complication was not seen in any of the cases in the study by Yeo et al. [
8]. This condition might occur because of ciliary body atrophy or detachment due to the cyclitic membrane, and it developed in six eyes in our study (2.6%), five of which likely resulted from incomplete retinal attachment. In one case, ocular hypotony led to phthisis bulbi. Band keratopathy, a known complication of SO, was seen in five eyes (2.2%) in our series and has been reported to occur in 3% [
15] and 24% [
11] in diabetic eyes that underwent vitrectomy plus SO injection. Other complications noted in this study are not very common and so might not have been very influential.
One limitation was the use of 20-gauge instruments for all the operations; these tools are currently not commonly used by many surgeons. This study was retrospective; however, it is powered by a relatively large sample size and long term follow-up. The results indicated that, although anatomical outcomes were acceptable in eyes with advanced diabetic retinopathy treated by vitrectomy plus SO injection, the functional prognosis was not that satisfying. This unfavorable visual outcome is related mostly to the nature of the disease rather than to the effects of the surgery or SO complications. However, the observed relatively poor visual results in this study should not discourage consideration of this procedure for advanced cases of diabetic retinopathy, since the mean BCVA improved significantly postoperatively and persisted for a long period of time. In addition, our result emphasized that retention of SO in the eye in selected cases and for an extended period of time would not affect the outcome.
The results of this study will help both retinal surgeons and diabetic patients to decide whether to undergo advanced surgical intervention. Our findings provide physicians and their patients a practical expectation of the functional and anatomical outcomes, as well as the likelihood for reoperation, the potential postoperative complications, and the various cares needed after the surgery. However, complementary studies using smaller gauge vitrectomies and more delicate instruments are recommended.








 PDF Links
PDF Links PubReader
PubReader Full text via DOI
Full text via DOI Full text via PMC
Full text via PMC Download Citation
Download Citation Print
Print



