 |
 |
| Korean J Ophthalmol > Volume 28(4); 2014 > Article |
Abstract
Purpose
To investigate patterns of subsequent progression of localized retinal nerve fiber layer (RNFL) defects and to quantify the extent of progression in normal-tension glaucoma (NTG) patients.
Methods
Thirty-three eyes of 33 consecutive NTG patients who had shown continuous progression of localized RNFL defect on serial red-free fundus photographs were selected for the study. Patterns of subsequent progression of localized RNFL defects were categorized, and extents of progression were quantified. Serial evaluations of disc stereophotographs and visual fields were also performed to detect progression.
Results
The most common pattern was continuous widening of the defect towards the macula (n = 11, 33.3%) followed by sharpening of the defect border after widening of the defect towards the macula (n = 5, 15.2%), continuous widening of the defect away from the macula (n = 2, 6.1%), and deepening of the defect after appearance of a new defect (n = 2, 6.1%). Four eyes (12.1%) simultaneously showed two patterns of subsequent progression. In 13 eyes that showed continuous widening of the defect, subsequent angular widening towards the macula and away from the macula were 9.2 ┬▒ 6.0┬░ (range, 1.1┬░ to 24.4┬░; n = 11) and 5.2 ┬▒ 4.9┬░ (range, 0.3┬░ to 11.3┬░; n = 2), respectively. Thirty-two eyes showed no progression of optic disc cupping. Out of the 21 eyes in which Humphrey central 30-2 threshold visual field tests were performed after progression of RNFL defects, 15 eyes showed no deterioration in the visual field.
Conclusions
There were nine patterns of subsequent progression of localized RNFL defects. Among them, continuous RNFL loss proceeding temporally was the most common one. Initial progression of the defect proceeded temporally, especially in the defect located at the inferior fundus, might be at a risk of further RNFL loss temporally.
The glaucomatous damage is visible both in the retinal nerve fiber layer (RNFL) and the optic disc [1]. RNFL defect is the most sensitive parameter in detecting glaucomatous optic neuropathy [2], and serial RNFL examinations by red-free fundus photography are known to be more sensitive than color disc photographs or visual fields evaluations in detecting progression of glaucomatous optic neuropathies [3,4]. As the differences among optical coherence tomography (OCT) devices with regard to the measurement protocols and thickness values affect their ability to detect localized RNFL defects, red-free fundus photography still remains a useful and complementary tool in detecting RNFL defects and monitoring the progression of the defects. In the detection of localized RNFL defects using OCT devices, the deviation-from-normal map of the Cirrus OCT device and the 36-segment map of the 3D OCT device have the highest sensitivity (89.6% and 91.7%, respectively) compared to the circumpapillary RNFL parameters of both devices [5]. The sensitivity of the Spectralis OCT parameters ranged from 18.9% to 83.8% [6]. Furthermore, the Spectralis HRA+OCT device revealed various degrees of circumpapillary RNFL thinning and disruption of circumpapillary RNFL reflectivity in the same location as the photographic RNFL defect for 23 (35.4%) of the 65 RNFL defects [7]. An earlier study evaluated progression of localized RNFL defects on red-free fundus photographs in normal-tension glaucoma (NTG). In most cases, RNFL loss proceeded temporally [8]. But little is known about patterns of subsequent progression of RNFL defects after initial progression. We investigated patterns of subsequent progression of localized RNFL defects on serial red-free fundus photographs and quantified extents of progression in NTG patients. This study may provide us which pattern of initial progression of localized RNFL defect is at a risk of further progression of defect.
Thirty-three eyes of 33 consecutive NTG patients that had shown subsequent progression of localized RNFL defects, as revealed by serial red-free fundus photographs, were selected by one ophthalmologist (DMK) for the study. These patients were followed up at the Glaucoma Clinic of Seoul National University Hospital between March 2001 and July 2013. NTG was defined as diurnal intraocular pressure (IOP) of 21 mmHg or lower without any medication, glaucomatous optic neuropathy, glaucomatous visual field defect, open anterior chamber angle, and no evidence of non-glaucomatous cause of optic nerve damage. Each patient had measurement of visual acuity, refraction, slit-lamp biomicroscopy, diurnal IOP measurements with Goldmann applanation tonometer, central corneal thickness measurement, gonioscopy, disc stereophotography, red-free fundus photography, and automated perimetry. When the result of Humphrey central 30-2 threshold visual field test was within normal limits (n = 10), Humphrey Matrix 30-2 visual field test was performed. Disc stereophotography, red-free fundus photography, and automated perimetry were performed annually or between 6 months and one year after the previous examination. Disc stereophotograph and red-free fundus photograph were taken with a digital fundus camera (VX-10; Kowa, Tokyo, Japan) after full dilation of the pupil. Sixty-degree, wide-angle views of the optic disc, carefully focused on the retina using the built-in split-line focusing device and centered between the fovea and the optic disc, were obtained for red-free fundus photographs and were reviewed on a LCD monitor.
Localized RNFL defect was defined as a dark wedge-shaped area with its tip touching the optic disc margin in the brightly striated pattern of the surrounding RNFL. The width of the defect at 1-disc diameter distance from the margin of the optic disc should be larger than that of the major retinal vessel [9]. The parameters, angle ╬▒ and angle ╬▓, used to represent localized RNFL defects were defined as described elsewhere [8]. Angle ╬▒ is the minimum angle that is made by a line from the center of the optic disc to the center of the macula and a line from the center of the optic disc to the optic disc margin where the nearest border of the RNFL defect meets the optic disc. The smaller the angle, the closer the RNFL defect to the fovea and the closer the visual field defect to the fixation. Angle ╬▓ is the circumferential angle of the RNFL defect. The larger the angle, the wider the RNFL defect in circumferential width and the more diffuse the visual field defect.
Two examiners (TJK and YKK) masked to the subjects' identities and test results classified 33 eyes into the following categories after reviewing serial red-free fundus photographs: widening or deepening of the pre-existing defect, appearance of a new defect, or an ambiguous one. Widening was defined as a recognizable change in the distance between the border of the RNFL defect and the adjacent reference vessel. In these cases, the difference of angle ╬▓ and the direction of the widening (towards or away from the macula) were determined using the angle-measuring program (Maro-view 5.4; Infinitt, Seoul, Korea) on a LCD monitor. Depth of the RNFL defect was determined according to the following two methods after equalizing brightness and contrast of RNFL at the posterior pole among images on the LCD monitor. First, severity of the RNFL defect was divided into three grades (grade 1, mild defect; grade 2, moderate defect; grade 3, severe defect) by evaluating brightness and texture of the RNFL and degree of blood vessel obscuration. This method was described in detail elsewhere [2]. Grade 1 indicates fine striations of RNFL are visible and only large vessels are clearly visible; grade 2, striations of RNFL are barely detectable and even small vessels are clearly visible; and grade 3, texture of the RNFL is not visible at all. Second, sharpness of temporal and nasal borders of the RNFL defect was compared among images. Deepening was defined as an increase in the grade of severity or as sharpening of the border of the pre-existing RNFL defect. Appearance of a new defect was determined when a localized defect ranging from grade 1 to 3 developed in the area where there had been no previous defect [2]. When the photograph had poor image quality or a change was too subtle to be considered progression, these cases were classified as ambiguous one and excluded from further evaluation. Evaluation of serial red-free fundus photographs was independently performed twice by each examiner (TJK and YKK) and inter-observer discrepancies were resolved by adjudication of a third experienced examiner (DMK). Absolute values and changes of angle ╬▒ and angle ╬▓ were determined by averaging values measured by the two examiners. In the case of bilateral progression, only one eye was randomly selected.
Evaluations of serial disc stereophotographs and visual fields were also performed to detect progression. Progressive changes of the optic disc were defined as narrowing or notching of the neural rim, increased cup:disc ratio, or shift in the position of the vasculature. The optic disc changes were also evaluated by the two examiners in the same manner as done in the evaluation of red-free fundus photographs and the discrepancy between them was resolved by adjudication of a third experienced examiner. Visual field defect progression was defined according to the GPA program of the Humphrey Field Analyzer. In cases that showed progression on the visual fields, pattern of progression was divided into three categories: deepening or extension of the pre-existing scotoma or a new scotoma not within or adjacent to the pre-existing scotoma [10]. The study protocol was approved by the institutional review board of Seoul National University Hospital Biomedical Research Institute (no. 1309-008-516). Statistical analysis was performed using MedCalc ver. 12.7.0 (MedCalc, Ostend, Belgium).
Among the 33 eyes of the 33 NTG patients enrolled, no eye was classified as ambiguous one or as showing bilateral continuous progression. Demographics and ophthalmic measurements are presented in Table 1. Mean age at diagnosis was 54.2 ┬▒ 11.4 years, and female patient comprised 57.6% of the study group. Mean follow-up period was 52.5 ┬▒ 31.5 months (range, 16 to 131 months). No patient had intraocular surgery other than cataract surgery or laser treatment. There was strong intra- and inter-observer agreements in determining progression of the RNFL defect for each progression criterion (widening of the defect towards the macula or away from the macula, severity grading of the defect, sharpening of the defect border, and occurrence of a new defect) (k-values Ōēź0.80).
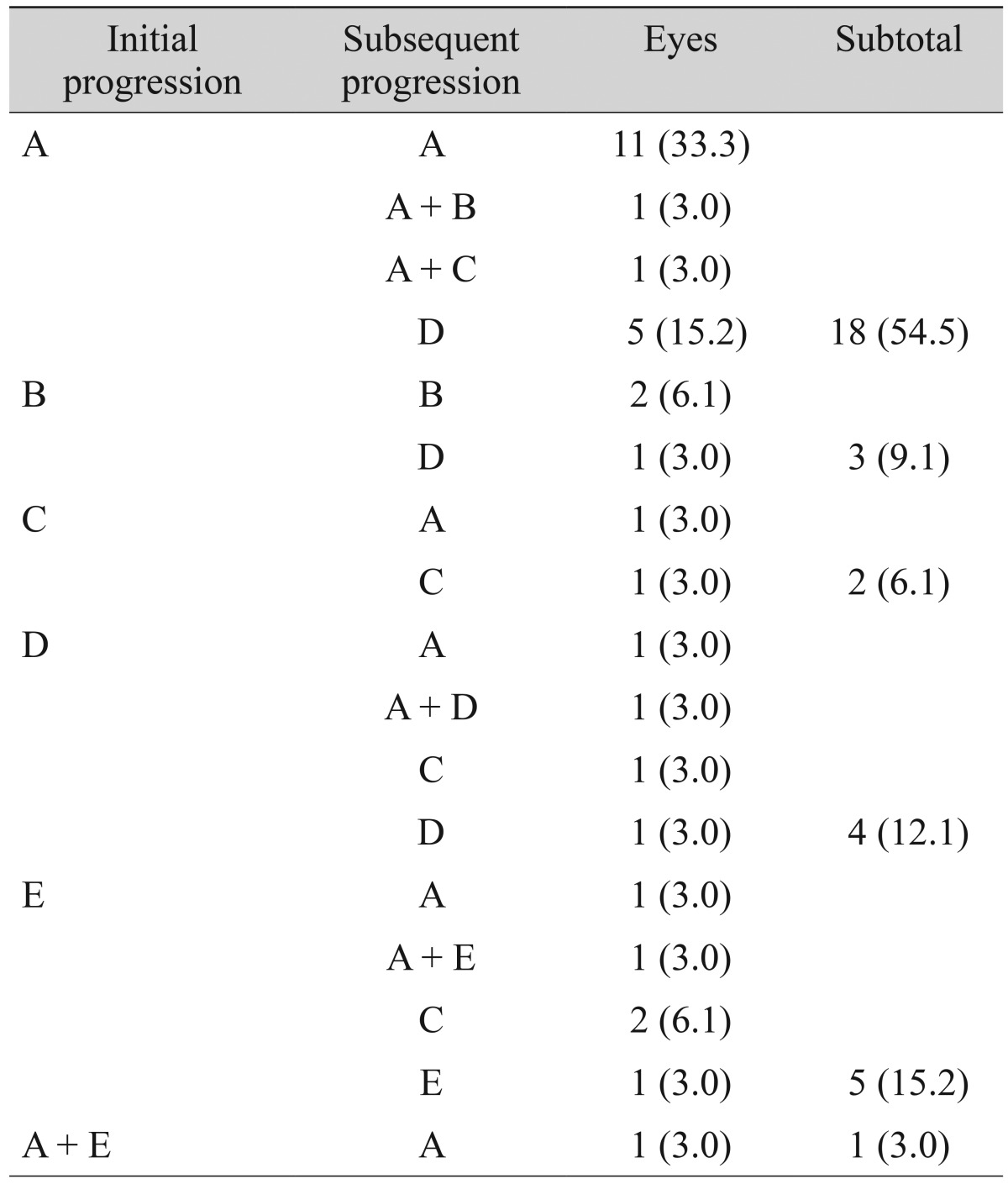
At the initial progression of the RNFL defect, the most common pattern of progression was widening of the defect towards the macula (54.5%) followed by appearance of a new defect (15.2%), sharpening of the defect border (12.1%), widening of the defect away from the macula (9.1%), and deepening of the defect (6.1%). One eye simultaneously showed two patterns of progression which were widening of the defect towards the macula and appearance of a new defect (Table 2).
Regarding subsequent progression of RNFL defect, most of localized RNFL defects were located at both of the superotemporal and inferotemporal retina (n = 16). The most common pattern was continuous widening of the defect towards the macula (n = 11, 33.3%) followed by sharpening of the defect border after widening of the defect towards the macula (n = 5, 15.2%), continuous widening of the defect away from the macula (n = 2, 6.1%), and deepening of the defect after appearance of a new defect (n = 2, 6.1%) (Fig. 1). Four eyes (12.1%) simultaneously showed two patterns of subsequent progression. They had the common feature of widening of the defect towards the macula. Other combined patterns were widening of the defect away from the macula, deepening of the RNFL defect, sharpening of RNFL defect border, and appearance of a new defect, respectively, in each eye (Table 2). In 13 eyes that showed continuous widening of the defect, subsequent angular widening towards the macula and away from the macula were 9.2 ┬▒ 6.0┬░ (range, 1.1┬░ to 24.4┬░; n = 11) and 5.2 ┬▒ 4.9┬░ (range, 0.3┬░ to 11.3┬░; n = 2), respectively. In five eyes that showed deepening of the defect, severity of the defect was increased by one grade from grade 1. Only one eye showed continuous deepening of the defect. Ten eyes had sharpening of the defect border at initial or subsequent progression, and the location was mostly at the temporal border (n = 8). Continuous widening of the defect towards the macula occurred in 8 eyes (44.4%) out of 18 eyes with defects located at the inferior fundus, and in 3 eyes (20%) out of 15 eyes with defects located at the superior fundus.
Thirty-two eyes showed no progression of optic disc cupping. One eye showed increased notching at the time when initial progression of the RNFL defect was detected. Optic disc hemorrhages were found on the disc stereophotographs taken just before the detection of continuous progression of the RNFL defect (n = 7), at the time of detection (n = 1), and at both (n = 1).
Out of the 21 eyes in which Humphrey central 30-2 threshold visual field tests were performed after progression of RNFL defect, 15 eyes showed no deterioration in the visual field and 1 eye was categorized as having a visual field with low reliability. Three eyes showed 'likely progression' involving deepening (n = 2) and extension (n = 1) of pre-existing scotoma. Two eyes showed 'possible progression' involving either deepening of pre-existing scotoma or a new scotoma. In the remaining 12 eyes, 6 eyes had Humphrey Matrix 30-2 visual field defect at the time of diagnosis and normal visual fields at the follow-up Humphrey central 30-2 threshold visual field tests, 2 eyes showed conversion of Humphrey Matrix 30-2 visual field defect to Humphrey central 30-2 threshold visual field defect at the follow-up, 1 eye had Humphrey Matrix 30-2 visual tests at the time of diagnosis and follow-up, and 3 eyes had no visual field tests performed at the time when progression of the RNFL defect was detected.
In this study, subsequent progression of localized RNFL defects was categorized into nine patterns. The most common pattern was continuous widening of the defect towards the macula (33.3%) followed by sharpening of the defect border after widening of the defect towards the macula (15.2%), continuous widening of the defect away from the macula (6.1%), and deepening of the defect after appearance of a new defect (6.1%). To our knowledge, no earlier study had categorized patterns of subsequent progression of localized RNFL defects. At the initial progression of RNFL defects, the most common pattern of progression was widening of the defect towards the macula (54.5%) followed by appearance of a new defect (15.2%), sharpening of the defect border (12.1%), widening of the defect away from the macula (9.1%), and deepening of the defect (6.1%). Suh et al. [8] reported that the most common pattern of progression was widening of the defect towards the macula (56.9%) followed by deepening of the defect (38.5%), appearance of a new defect (9.2%), and widening of the defect away from the macula (7.7%) in NTG patients who showed progression of localized RNFL defects for the first time. These findings suggest that widening of the defect towards the macula for the initial progression might be at a risk of further progression of the RNFL defect. Sharpening of the defect border mostly developed at the temporal border. This is a characteristic feature of deepening of the RNFL defect. As retinal nerve fibers are less detectable in the nasal fundus than in the temporal fundus, RNFL loss should proceed temporally [11]. This finding concurs with the earlier report suggesting that the border of the RNFL defect is the most active site of loss of retinal ganglion cells and their axons [12].
This study shows that subtle progression of localized RNFL defects can be detected by serial red-free fundus photography, and the changes were hardly detected by disc stereophotography. Therefore, red-free fundus photography should have a role in the early detection of progression of glaucoma in addition to disc stereophotography. It is sometimes difficult to clearly define the border of diffuse atrophy using red-free fundus photography, and this may lead to problems in categorizing patterns of progression [13]. Therefore, this study only included localized defects. There remains a question as to whether deepening of the RNFL defect can be detected objectively. However, this study showed good intra- and inter-observer agreement in grading severity of the defect and determining sharpening of the defect border. It should be noted that the study is inherently limited by not applying OCT. In spectral-domain OCT, guided progression analysis was introduced in 2009 (ver. 4.0) to facilitate topographic analysis of RNFL progression. By tracking RNFL changes with serial, registered RNFL thickness maps, GPA compares RNFL thickness of individual pixels between the baseline and follow-up images and provides a visual display of the area and location of significant change. However, the Cirrus OCT deviation-from-normal map had the lowest cutoff angle for the width (10.69┬░) of the localized RNFL defect [14]. In 13 eyes that showed continuous widening of the defect, subsequent angular widening towards the macula and away from the macula were 9.2 ┬▒ 6.0┬░ and 5.2 ┬▒ 4.9┬░, respectively. Further longitudinal studies are warranted to elucidate the changes and to compare detection of defect progression between red-free fundus photography and OCT.
In conclusion, there were nine patterns of subsequent progression of localized RNFL defects. Among them, continuous RNFL loss proceeding temporally was the most common one. Initial progression of defects proceeded temporally, especially in the defect located at the inferior fundus, might be at a risk of further RNFL loss temporally.
REFERENCES
1. Tuulonen A, Airaksinen PJ. Initial glaucomatous optic disk and retinal nerve fiber layer abnormalities and their progression. Am J Ophthalmol 1991;111:485-490.


2. Quigley HA, Reacher M, Katz J, et al. Quantitative grading of nerve fiber layer photographs. Ophthalmology 1993;100:1800-1807.


3. Sommer A, Miller NR, Pollack I, et al. The nerve fiber layer in the diagnosis of glaucoma. Arch Ophthalmol 1977;95:2149-2156.


4. Quigley HA, Katz J, Derick RJ, et al. An evaluation of optic disc and nerve fiber layer examinations in monitoring progression of early glaucoma damage. Ophthalmology 1992;99:19-28.


5. Kim KE, Ahn SJ, Kim DM. Comparison of two different spectral domain optical coherence tomography devices in the detection of localized retinal nerve fiber layer defects. Jpn J Ophthalmol 2013;57:347-358.


6. Jeoung JW, Kim TW, Weinreb RN, et al. Diagnostic ability of spectral-domain versus time-domain optical coherence tomography in preperimetric glaucoma. J Glaucoma 2013 1 31 http://dx.doi.org/10.1097/IJG.0b013e3182741cc4.

7. Nukada M, Hangai M, Mori S, et al. Detection of localized retinal nerve fiber layer defects in glaucoma using enhanced spectral-domain optical coherence tomography. Ophthalmology 2011;118:1038-1048.


8. Suh MH, Kim DM, Kim YK, et al. Patterns of progression of localized retinal nerve fibre layer defect on red-free fundus photographs in normal-tension glaucoma. Eye (Lond) 2010;24:857-863.


9. Hoyt WF, Frisen L, Newman NM. Fundoscopy of nerve fiber layer defects in glaucoma. Invest Ophthalmol 1973;12:814-829.

10. Boden C, Blumenthal EZ, Pascual J, et al. Patterns of glaucomatous visual field progression identified by three progression criteria. Am J Ophthalmol 2004;138:1029-1036.


11. Jonas JB, Schiro D. Localised wedge shaped defects of the retinal nerve fibre layer in glaucoma. Br J Ophthalmol 1994;78:285-290.



12. Ishida K, Yamamoto T, Sugiyama K, Kitazawa Y. Disk hemorrhage is a significantly negative prognostic factor in normal-tension glaucoma. Am J Ophthalmol 2000;129:707-714.


13. Airaksinen PJ, Tuulonen A, Werner EB. Clinical evaluation of the optic disc and retinal nerve fiber layer. In: Ritch R, Shields MB, Krupin T, The glaucomas. 2nd ed. St Louis: Mosby; 1996. p. 617-657.
Fig.┬Ā1
Red-free fundus photographs show patterns of continuous progression of localized retinal nerve fiber layer defects such as (A) continuous widening (arrow heads and empty arrow) of the defect towards the macula, (B) continuous widening (arrow heads and empty arrow) of the defect away from the macula, (C) continuous sharpening of the border (arrow heads and empty arrow) of the defect, (D) continuous appearance of a new defect (arrow heads and empty arrow), and (E) appearance of a new defect (arrow heads) followed by deepening of the defect (empty arrow). Arrows in the left first photographs indicate the location of the defect.

Table┬Ā1
Demographics and ophthalmic measurements in the eyes with continuous progression of localized RNFL defects (n = 33)




 PDF Links
PDF Links PubReader
PubReader Full text via DOI
Full text via DOI Full text via PMC
Full text via PMC Download Citation
Download Citation Print
Print




