 |
 |
| Korean J Ophthalmol > Volume 28(3); 2014 > Article |
Abstract
Purpose
Since few reports had been published on the prevalence of toxocariasis in ankylosing spondylitis (AS) patients with acute non-granulomatous anterior uveitis (ANGAU), the aim of this work was to determine the presence of antibodies against Toxocara canis in AS patients with ANGAU.
Methods
Thirty-six patients (14 female and 22 male) with AS were enrolled in the study. The history of ANGAU was accepted only if diagnosed by an ophthalmologist. The detection of IgG antibodies to T. canis was determined by enzyme-linked immunosorbent assay. In addition, antibodies to Ascaris lumbricoides were also tested to verify non-specific reactions.
Results
The prevalence of ANGAU in the AS patients was 58% (21 / 36), and 38% (8 / 21) of the patients with ANGAU were positive for antibodies to Toxocara, while 7% (1 / 15) of AS patients without ANGAU were positive for T. canis (p = 0.038, two tails; mid-p exact). No antibodies were detected to A. lumbricoides antigens in the serum samples of patients with AS.
Conclusions
These data suggest that the seroprevalence of antibodies to T. canis is high in Mexican patients with AS-associated uveitis, suggesting a chronic asymptomatic toxocariosis, which could be associated with the pathogenesis of ANGAU; however, further larger-scale studies are needed to confirm this observation.
Acute anterior uveitis is the most common form of uveitis and is also a common cause of vision loss in most regions of the world, but the exact etiology cannot be elucidated in some cases. Uveitis is a sight-threatening inflammation inside the eye that affects both the uveal tract and adjacent structures and is the most frequent extra-articular manifestation of ankylosing spondylitis (AS). The prevalence of acute non-granulomatous anterior uveitis (ANGAU) in AS patients has been reported to be as high as 25% to 40% during the course of the rheumatic disease [1,2,3]. In our experience, only 2% of patients are accurately diagnosed by an ophthalmologist at the onset of AS, although 22% of chronic AS patients develop uveitis, thus the availability of correctly characterized biological samples for further analysis is limited. The pathogenesis of AS with ANGAU usually has a strong genetic linkage with human leukocyte antigen (HLA)-B27 [4]. In addition, a genetic association of HLA with human infection by helminthes has been reported [5].
Infection with Toxocara canis is among the most common zoonotic infections worldwide and is a well-recognized cause of uveitis worldwide [6,7]. Humans can be infected with these parasites by ingestion of soil or contaminated meat containing Toxocara eggs. Ocular toxocariasis causes permanent vision loss in many patients and Toxocara is an important causative agent of posterior and diffuse uveitis [7]. However, Toxocara should be considered as a possible causative agent of ANGAU. There is some evidence that the occurrence of helminthes and the prognosis of rheumatic disease are linked [8,9]. In this context, Peng [10] has reported that potential parasitosis must be considered in patients with rheumatic diseases. Williams and Roy [11] have reported a case of arthritis associated with toxocaral infestation. Furthermore, an association of arthritis with infection due to helminthes, such as Strongyloides stercoralis, has been reported [12,13]. A study conducted by Kuk et al. [14] reported Toxocara seropositivity in AS patients.
Reports regarding the influence of toxocariasis in patients with AS-associated uveitis are limited and the relationship between toxocariasis and AS-associated uveitis is unclear. Thus, the aim of this work was to determine the presence of antibodies against T. canis in AS patients with ANGAU. To our knowledge, this is the first report showing the relationship between Toxocara and Mexican AS patients with uveitis.
Thirty-six AS patients (14 female and 22 male; mean ┬▒ standard deviation [SD] age, 39.7 ┬▒ 15.1 years) participated during the study period. We only included patients residing in Mexico City. Twenty-one of the 36 patients showed acute anterior uveitis, so they were first diagnosed by an ophthalmologist and then submitted to rheumatology to complete their diagnosis and treatment. No symptoms or signs of Toxocara infection were identified at this point. Initial assessment included collection of demographic information by questionnaire. During the appointment, each patient was invited to participate in this study. The mean ┬▒ SD age at onset of AS was 24.6 ┬▒ 11.6 years and disease duration was 14.6 ┬▒ 13.6 years. HLA-B27 was positive in 62.5% (20 / 32) of AS patients (Table 1), but no data were available in four patients without uveitis. All AS patients fulfilled the 1984 modified New York criteria for diagnosis of AS [15] and completed questionnaires assessing functional ability (BASFI [Bath Ankylosing Spondylitis Functional Index]). This questionnaire includes 10 questions; eight evaluate activities related to the condition of the spine and two questions evaluate the patient's ability to cope with daily life. Those patients with other spondyloarthropathies or hepatitis B or C were excluded. The control group was formed from 10 samples from healthy individuals as well as 10 samples from patients with toxocariosis and 10 with ascariasis.
The presence of IgG antibodies directed to T. canis or Ascaris lumbricoides was determined by enzyme-linked immunosorbent assay (ELISA) as previously reported [16]. To this end, blood samples (10 to 15 mL) were collected and separated serum samples were stored at -20Ōäā until used. Excretory and secretory antigens of T. canis adult worms as well as a crude extract of A. lumbricoides adult worms were prepared. Worms were obtained from natural infections. The T. canis or the A. lumbricoides antigen was diluted in 100 mM carbonate-bicarbonate buffer, pH 9.6. Flat-bottom polystyrene plates (Corning-Costar, Tewksbury, MA, USA) were coated at 100 ┬ĄL/well with the antigen solution, incubated overnight at 4Ōäā, and then washed three times with 0.01 M phosphate-buffered 0.15-M saline (PBS) pH 7.2 containing 0.05% Tween 20 (PBS-T). Wells were blocked with 1% nonfat milk for 2 hours at 37Ōäā, and were washed with PBS-T. Individual serum samples of 100 ┬ĄL were added to the wells in triplicate, incubated for 2 hours at 37Ōäā, and washed again. Anti-human IgG-horseradish peroxidase conjugate (Zymed-Invitrogen, Camarillo, CA, USA) diluted 1 : 4,000 in PBS-T was added, and incubated for 2 hours at 37Ōäā. After washing, a substrate reaction mixture solution prepared with o-phenylenediamine (Sigma-Aldrich, St. Louis, MO, USA) and H2O2 (Merck KGaA, Darmstadt, Germany) dissolved in 75 mM phosphate-citrate buffer, pH 5, was added to the wells. The enzyme reaction was terminated with 2 M H2SO4, and absorbency values were determined with an ELISA reader at 490 nm. The cutoff value was previously calculated [16,17] as the mean ┬▒ 3 times the SD of 77 serum samples obtained from healthy people (31 male and 46 female; age mean ┬▒ SD, 30 ┬▒ 15 years) and was used routinely to perform the diagnosis in clinical cases. In this study, to validate the results of the ELISA, 10 samples from healthy individuals were used as the negative control, and 10 samples of patients with ascariasis and 10 with toxocariasis were used as positive controls.
Chi-square and the mid-p exact tests as well as the odds ratio (OR) and their 95% confidence intervals (CI) between patients with and without ANGAU were used to analyze group differences. Otherwise, statistical analysis was performed using the statistical package Epi-Epidemiology ver. 2 (http://www.openepi.com). A p-value <0.05 was considered significant.
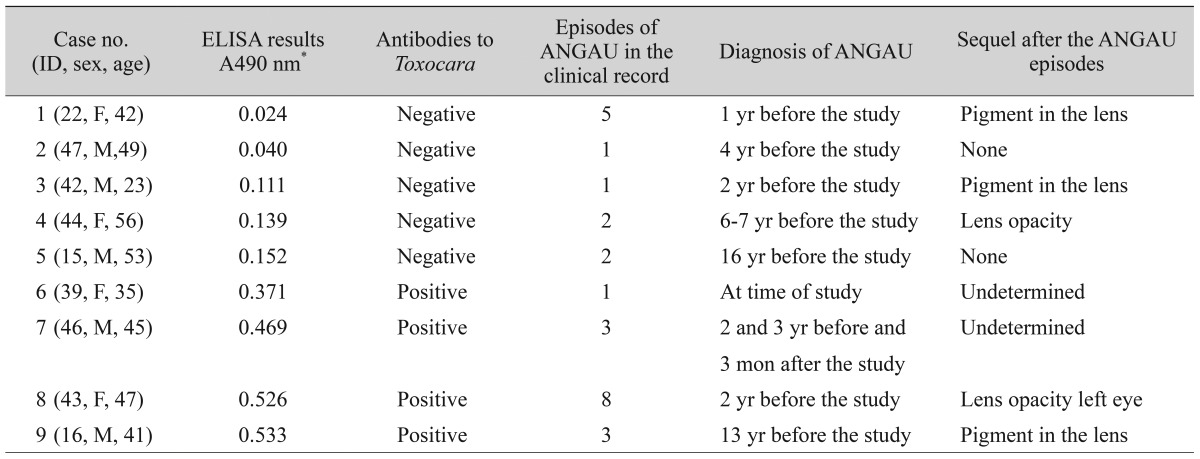
Table 1 shows the demographics and clinical characteristics of the 36 AS patients. The ages of the patients ranged from 16 to 66 years (mean ┬▒ SD, 39.7 ┬▒ 15.1 years). The prevalence of ANGAU in the AS patients was 58% (21 / 36); eight (38%) of the AS patients with ANGAU were female and 13 (62%) were male and the ages of these patients ranged from 16 to 65 years (age mean ┬▒ SD, 37.9 ┬▒ 14.5 years). Regarding the HLA-B27 test, 63.2% (12 / 19, no data were available in three cases) of AS patients with ANGAU were positive, while 57.1% (8 / 14, no data was available in one case) of AS patients without ANGAU had positive HLA-B27 (p = 0.4311, chi-square test, two tails; OR, 1.103; 95% CI, 0.3117 to 4.024). According to the available data, a total of 26 episodes of ANGAU were recorded in the AS patients with a mean of 2.89 ┬▒ 2.31 (range, 1 to 8) episodes by patient (Table 1). The number of episodes of ANGAU and clinical features of the involved eyes are presented in Table 2.
AS patients with ANGAU had significantly higher absorbance values of antibodies against T. canis (0.228 ┬▒ 0.191) than those without ANGAU (0.111 ┬▒ 0.107; p = 0.026; Student's t-test, 2 tails) (Table 1). By contrast, there was not a statistically significant difference between AS patients with and without ANGAU in the absorbance values of antibodies against A. lumbricoides, being 0.079 ┬▒ 0.109 and 0.112 ┬▒ 0.089 (p = 0.428) respectively.
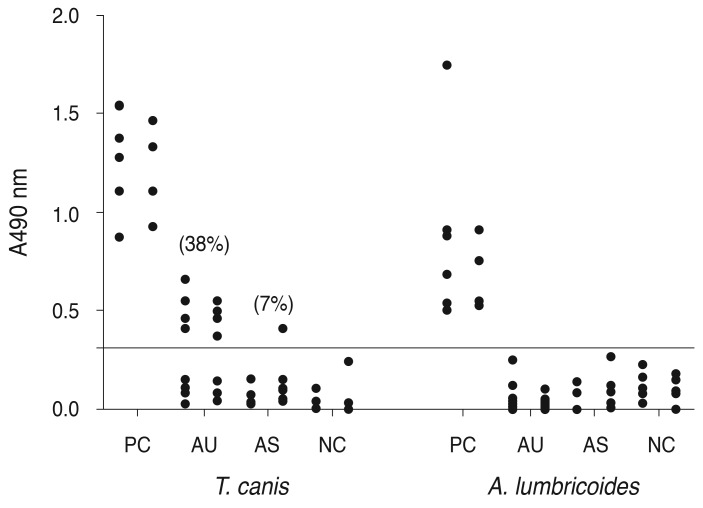
Individual results showed that 38% (8 / 21) of the patients with ANGAU were positive for antibodies to Toxocara, while 7% (1 / 14) of AS patients without ANGAU had positive serum samples for antibodies to Toxocara. A chi-square test was performed between the condition of ANGAU and the presence of antibodies to T. canis (p = 0.032). However, since at least one expected value in the contingence table was <5, the mid-p exact test was considered rather than the chi-square test (p = 0.019; OR, 8.174; 95% CI, 1.095 to 205.3). In four patients with AS and positive serology against T. canis, one had an ANGAU episode 13 years before our study, and in another patient the ANGAU episode occurred a year before our study, while yet a different patient had ANGAU at the time of the study and another one had a relapse of ANGAU which occurred three months later (Table 2). In addition, the frequency of serum samples with antibodies against T. canis was significantly higher in AS patients with ANGAU than in the healthy control individuals (Fig. 1).
The occurrence of ANGAU in our group of patients was as high as 58% during the course of the disease. This correlates with previous data that found that the prevalence of ANGAU in AS is common during the course of the disease [2]. However, the association of toxocariasis in AS patients with ANGAU is uncertain. This is the first report that specifically highlighted the prevalence of T. canis infection in AS patients with ANGAU. Our results indicate that AS patients with ANGAU had a significantly higher prevalence of T. canis than those without. On the contrary, there was no difference in seroprevalence against A. lumbricoides between AS patients with and without ANGAU, suggesting specificity of the antibodies to T. canis and their possible involvement in the pathogenesis of ANGAU. Although the existence of cross-reactivity among antigens of T. canis and A. lumbricoides has been reported, there exist specific antigens of T. canis that enables its diagnosis. In fact, the detection of antibodies against T. canis has been used to confirm toxocariasis [18,19]. While a Western blotting procedure could be more suitable for the serologic diagnosis of toxocariasis in Mexican patients, the sensitivity of the ELISA technique has been particularly recommended in the diagnosis of toxocariasis in seroepidemiological studies [16,20,21].
It has been reported that human toxocariasis is found in many countries around the world and, prevalence rates can reach as high as 40%. In Mexico, data regarding prevalence and distribution of toxocariasis are scarce. Some studies along the Mexico-United States border region reported 10.6% of seroprevalence [22]; however, the potential of infection is very high in metropolitan areas in Central Mexico, since the soils in public parks are highly contaminated (30.3%), as well as dog feces found in soil (28.1%) or samples from dogs with owners (39.8%) [23]. Geographic location plays a role in the epidemiology of Toxocara because it is more prevalent in hot, humid regions where eggs are kept viable in the soil, as demonstrated by the following data, where the prevalence of toxocariosis has been recorded by geographic location: Argentina (the prevalence of toxocariasis in rural locations was of 23% to 31.6%; whereas, the prevalence of toxocariasis in urban localities was of 37.9% to 46.9%), Bolivia (rural 27%), Brazil (rural 13.7%, urban 21.5%), Peru (rural 44.9%, urban 7.3%), Colombia (urban 47.5%), Cuba (rural 57.5%, urban 5.2%), Trinidad y Tobago (62.3%), Mexico (urban 7.5%), USA (13.9%), Kenya (rural 7.5%), Nigeria (urban 30.4%), Indonesia (rural 84.6%, urban 12.2%), Iran (25.6%), Jordan (10.9%), Korea (rural 5%), Taiwan (rural 76.6%), Denmark (urban 2.4%), Ireland (rural 31%), Poland (urban 20.7%), and Spain (urban 28.6% to 65.7%) [24,25].
There are several factors that have been associated with higher rates of infection with Toxocara. People are more likely to be infected with Toxocara if they own a dog. A number of studies worldwide have estimated that there is one dog for every 10th person and, in many countries around 50% are stray dogs. In Mexico, the frequency of dogs is estimated to be 1 dog for every 3rd person. Recently the prevalence of anti-T. canis antibodies in stray dogs in Mexico City was reported, and results showed that from 141 sera, 94 (40 males and 54 females) proved to have a positive reaction with a global infection prevalence of 66.7% [26]. Therefore, having a litter of puppies in the home has been identified as a significant risk factor [6]. Many of the AS patients studied here were pet owners at the time of the study. Our results found that 62% of the patients with ANGAU were male, and 38% were female. This is in agreement with a previous study that showed that the demographic characteristics of patients with uveitis due to Toxocara have been reported to be fewer in female than in male patients [27]. However, we did not find an association of HLA-B27 positivity with patients with AS-associated uveitis. These results are in contrast with earlier findings indicating that HLA-B27-positive rates are significantly higher in AS patients with ANGAU that those without [28,29,30,31]. In addition, we did not a find correlation between the number of episodes of ANGAU, the clinical features of the involved eyes and the presence of antibodies to Toxocara (Table 2).
Although our results need to be reconfirmed by a larger-scale study, this data indicated that AS patients with a history of ANGAU had higher prevalence of toxocariasis, suggesting that the involvement of T. canis infection in the disease processes of AS in Mexican patients can lead to the development of ANGAU. Our data may represent an important report, since a high incidence of toxocariasis is found in Mexico. In addition, the dog population in Mexico is large, and most of our patients with active AS are dog owners; therefore, the lifestyle of these patients may constitute a risk factor for toxocariasis since they could be residentially exposed to pets. The information gained from this study may be of use for the implementation of an ophthalmological examination of patients seropositive to T. canis in public hospitals in Mexico.
Acknowledgements
Patricia Mendez-Samperio is a fellow in the programs of Stimuli Researchers Performance and, Operation and Development Committee of Academic Activities, National Politecnic Institute, in addition to the program of National System of Researchers, National Council of Science and Technology. Jorge-Luis de-la-Rosa-Arana is a fellow at the National System of Researchers, National Council of Science and Technology.
REFERENCES
1. Rosenbaum JT. Acute anterior uveitis and spondyloarthropathies. Rheum Dis Clin North Am 1992;18:143-151.


2. Gran JT, Skomsvoll JF. The outcome of ankylosing spondylitis: a study of 100 patients. Br J Rheumatol 1997;36:766-771.


3. Robertson LP, Davis MJ. A longitudinal study of disease activity and functional status in a hospital cohort of pa tients with ankylosing spondylitis. Rheumatology (Oxford) 2004;43:1565-1568.


5. Quinnell RJ. Genetics of susceptibility to human helminth infection. Int J Parasitol 2003;33:1219-1231.


6. Despommier D. Toxocariasis: clinical aspects, epidemiology, medical ecology, and molecular aspects. Clin Microbiol Rev 2003;16:265-272.



7. Rubinsky-Elefant G, Hirata CE, Yamamoto JH, Ferreira MU. Human toxocariasis: diagnosis, worldwide seroprevalences and clinical expression of the systemic and ocular forms. Ann Trop Med Parasitol 2010;104:3-23.


8. Bocanegra TS, Vasey FB. Musculoskeletal syndromes in parasitic diseases. Rheum Dis Clin North Am 1993;19:505-513.


9. Zaccone P, Fehervari Z, Phillips JM, et al. Parasitic worms and inflammatory diseases. Parasite Immunol 2006;28:515-523.



11. Williams D, Roy S. Arthritis and arthralgia associated with toxocaral infestation. Br Med J (Clin Res Ed) 1981;283:192.

12. Brocq O, Breuil V, Agopian V, et al. Reactive arthritis induced by Strongyloides stercoralis. Rev Rhum Engl Ed 1996;63:217-219.

13. Richter J, Muller-Stover I, Strothmeyer H, et al. Arthritis associated with Strongyloides stercoralis infection in HLA B-27-positive African. Parasitol Res 2006;99:706-707.


14. Kuk S, Ozgocmen S, Bulut S. Seroprevalance of toxocara antibodies in multiple sclerosis and ankylosing spondylitis. Indian J Med Sci 2006;60:297-299.


15. Van der Linden S, Valkenburg HA, Cats A. Evaluation of diagnostic criteria for ankylosing spondylitis: a proposal for modification of the New York criteria. Arthritis Rheum 1984;27:361-368.


16. Jimenez-Balderas FJ, Camargo-Coronel A, Gargia-Jaimes J, et al. A study on parasites in Mexican rheumatic disease patients. J Egypt Soc Parasitol 2012;42:271-280.


17. Altman DG. Practical statistics for medical research. London: Chapman and Hall; 1991. p. 229-276.
18. Embil JA, Tanner CE, Pereira LH, et al. Seroepidemiologic survey of Toxocara canis infection in urban and rural children. Public Health 1988;102:129-133.


19. Park SP, Park I, Park HY, et al. Five cases of ocular toxocariasis confirmed by serology. Korean J Parasitol 2000;38:267-273.



20. Elefant GR, Shimizu SH, Sanchez MC, et al. A serological follow-up of toxocariasis patients after chemotherapy based on the detection of IgG, IgA, and IgE antibodies by enzyme-linked immunosorbent assay. J Clin Lab Anal 2006;20:164-172.



21. De Visser L, Rothova A, de Boer JH, et al. Diagnosis of ocular toxocariasis by establishing intraocular antibody production. Am J Ophthalmol 2008;145:369-374.


22. Tinoco-Gracia L, Barreras-Serrano A, Lopez-Valencia G, et al. Seroprevalence of larva migrans of toxocara canis and evaluation of associated risk factors among children in a mexico-united states border region. Int J Appl Res Vet Med 2008;6:130-136.
23. Romero-Nunez C, Mendoza-Martinez GD, Bustamante LP, et al. Presence and viability of Toxocara spp in soils public parks, gardens and houses in dog feces Nezahualcoyotl, Mexico. Rev Cient 2011;21:195-201.
24. Romero N├║├▒ez C, Mendoza-Martinez GD, Yanez-Arteaga S, et al. Prevalence and risk factors associated with toxocara canis infection in children. ScientificWorldJournal 2013;2013:572089


25. Ponce-Macotela M, Martinez-Gordillo MN, Caballero Salazar S, et al. Toxocara and toxocariosis. In: Becerril-Flores MA, Medical parasitology. Mexico, M.F.: Mcgraw-Hill-Interamericana; 2004. p. 249-256.
26. Martinez-Barbabosa I, Quiroz MG, Gonzalez LA, et al. Prevalence of anti-T. canis antibodies in stray dogs in Mexico City. Vet Parasitol 2008;153:270-276.


27. Barisani-Asenbauer T, Maca SM, Hauff W, et al. Treatment of ocular toxocariasis with albendazole. J Ocul Pharmacol Ther 2001;17:287-294.


28. Khan MA, Kushner I, Braun WE. Comparison of clinical features in HLA-B27 positive and negative patients with ankylosing spondylitis. Arthritis Rheum 1977;20:909-912.


29. Jaakkola E, Herzberg I, Laiho K, et al. Finnish HLA studies confirm the increased risk conferred by HLA-B27 homozygosity in ankylosing spondylitis. Ann Rheum Dis 2006;65:775-780.


Fig.┬Ā1
Dispersion of optical densities (A490) obtained with serum samples from ankylosing spondylitis patients with (AU) and without (AS) a history of acute non-granulomatosus anterior uveitis. Detection of IgG serum antibodies against Toxocara canis and Ascaris lumbricoides was performed by enzyme-linked immunosorbent assay. Reactivity of serum from patients with toxocariosis and ascariasis is shown, respectively, in the positive control (PC) group. The samples of healthy donors were used as the negative control (NC). The horizontal line indicates the cutoff value.
Table┬Ā1
Demographic characteristics and the detection of antibodies against Toxocara canis and Ascaris lumbricoides in ankylosing spondylitis patients with and without a history of acute non-granulomatosus anterior uveitis

- TOOLS



 PDF Links
PDF Links PubReader
PubReader Full text via DOI
Full text via DOI Full text via PMC
Full text via PMC Download Citation
Download Citation Print
Print




