Modulation of Retinal Wound Healing by Systemically Administered Bone Marrow-Derived Mesenchymal Stem Cells
Article information
Abstract
Purpose
To evaluate whether systemically injected bone marrow-derived mesenchymal stem cells (MSCs) can be incorporated into neuroretinal tissues and play an important role in retinal wound healing in the laser-induced retinal trauma model.
Methods
Retinotomies were made by applying an Nd:YAG laser to rat retina. On the first day after the injuries, cell suspensions that were obtained from the same line of rat (containing 1 × 106 green fluorescence protein [GFP]-marked bone marrow-derived MSCs) were injected through a tail vein in the experimental group and phosphate buffer solution (PBS) was injected in the same way in the control group. Fundus photographs were taken serially for fundus examination and eyeballs were enucleated for histological studies that were conducted at five and seven weeks after MSC and PBS injection. After the tissues were prepared, the retinotomy sites were observed with routine histological staining and confocal microscopy.
Results
Retinal detachment resolved in the experimental group, whereas it progressed in the control group. The retinotomy sites closed partially with identifiable GFP positive cells 5 weeks after MSC injection. At 7 weeks after MSC injection, complete healing without retinal detachment and plentiful GFP positive cells were observed at the transitional zone between damaged and normal retina.
Conclusions
Systemically administered GFP-marked MSCs may be incorporated into the neuroretinal tissues and play an important role in the wound modulation of physically damaged retinal tissues.
Stem cells, featuring plasticity and self-renewal capacity, can differentiate into various types of cells and these characteristics offer huge potential for tissue regeneration and cell replacement therapy [1]. To date, several studies have shown that bone marrow cells differentiate into hepatocytes, vascular endothelial cells, epithelial cells, cardiac muscle cells, fibroblasts and various types of neural cells as well as hematopoietic stem cells. It has also been reported that bone marrow cells differentiate into many kinds of retinal cells [2-10]. Since bone marrow-derived mesenchymal stem cells (MSCs) are multipotent and their numbers can be expanded in culture, there has been much interest in their clinical potential for tissue repair and gene therapy. A number of studies have demonstrated the migration and multi-organ engraftment potential of MSCs in animal models and in human organ transplantations [1].
Retinas injury or weakened function will accordingly cause the permanent loss of visual activity [11]. Such diseases as age-related macular degeneration, glaucoma, retinitis pigmentosa and retinal detachment cause retinal degeneration in which various mechanisms are involved. In cases where retinal damage persists, the loss of visual activity will occur.
To treat these conditions, there have been many kinds of experimental attempts including drug therapy and the transplantation of sensory retina and electrical devices to stimulate the optic nerve or retinal neurons [12-14]. To stimulate neural transduction through the optic nerve and restore visual function in the neurosensory retina, many experimental attempts have been tried [12-14]. In the field of cell therapy, there have been many studies of retinal transplantation using newborn or embryonic stem cells, retina-derived stem cells, and hippocampus-derived neural stem cells, which have been shown to differentiate into neural-forming cells, which are then incorporated into the injured retina [15-18].
For the clinical application of stem cell transplantation into the retina, it is important that stem cells are transplanted into the retina efficiently. The intravenous injection of bone marrow cells into rats has been shown to induce neuronal differentiation in the brain [19]. However, whether or not bone marrow cells can target and incorporate into injured neuroretinal tissues after intravenous administration remains unclear.
In the present study, we investigate the feasibility of intravenously transplanted bone marrow-derived MSCs can target and incorporate into neuroretinal tissue in a laser-induced retinal injury model. Also, we evaluate whether MSCs have wound healing potential in the retinal injury model.
Materials and Methods
Materials
Eight 6-week-old Sprague-Dawley (SD) rats (Orient Bio, Seongnam, Korea) were used in this experiment. Four out of eight rats were injected with GFP-expressing MSCs after retinal damage was induced and the other animals were used as controls after laser treatment. All experiments were conducted in accordance with the ARVO statements for the Use of Animals in Ophthalmic and Vision Research.
Bone marrow harvest
Bone marrow-derived MSCs were extracted from six SD rats under deep anesthesia with phenobarbital sodium (10 mg/kg body weight, Jeil Pharm., Seoul, Korea). In each rat, the femur and tibia were isolated from the adjacent tissues, which were rinsed in 70% ethanol. Under sterile conditions, the edge of each bone was cut, Dulbecco's modified Eagle's medium (DMEM) was injected into the bone marrow using a 25-guage syringe, the bone marrow cells were pulled out to the opposite side, and this maneuver was repeated until the solution became transparent. The bone marrow cell extracts were centrifuged at 500 ×g for five minutes and then washed using new DMEM. Finally, cell suspensions were prepared using new DMEM containing 0.2% fetal bovine serum (FBS), 1% L-glutamate and 0.1% phosphate buffer solution (PBS).
Cultivation of bone marrow-derived MSCs
Prior to cell culture, cells were inoculated with DMEM with a high glucose content in which 10% FBS, 1% penicillin-streptomycin and 2% glutamine were added. Cells were then suspended at a concentration of 2 × 105/cm2 on a 75 cm2 tissue culture flask and were incubated at 37℃ in a 5% CO2 incubator. To make sure those cells were well engrafted, cultivating cells were incubated for 72 hours. Three rounds of washings were conducted in which non-adhesive cells were removed with medium replacement. Following this procedure, the culture medium was replaced at 4-day intervals. The cell concentration was maintained at 1 × 106 cells per flask.
Generation of retrovirus expressing green fluorescence protein
A retrovirus expressing green fluorescence protein (GFP) was generated as previously described [20].
Transduction of MSCs with GFP
Prepared MSCs were seeded in fibronectin-coated 6-well plates (Becton Dickison, San Jose, CA, USA) containing 5 µg/mL polybrene. MSCs were applied at a multiplicity of infection of 10. Cells were disclosed to viral supernatant, making a final volume of 3 mL in which the density was 1 × 106 cells per plate. Centrifugation was conducted at 800 ×g at 32℃ for 30 minutes. Then, an additional transduction was done for 48 hours. After washing with PBS to remove unabsorbed viral particles, trypsin was applied for five minutes. Puromycin was then added, by which GFP-transduced cells were selectively cultured.
Induction of retinal injury using the Nd:YAG laser
Pupils were dilated with 0.5% tropicamide and 2.5% phenylephrine. Two laser-induced retinal injuries were made in the right eye between the major vessels around the optic disc by a Nd:YAG laser (EPIC I; Coherent, Palo Alto, CA, USA) with an intensity of 2.0 to 3.0 mJ under anesthesia. Induction of full thickness retinal damage was confirmed with vitreous and subretinal hemorrhages.
Observations of fundus findings
The fundus changes were studied one day, three weeks, and five weeks after injury using fundus photography under anesthesia. The change over time in the findings of the vitreous and subretinal hemorrhages and the development of retinal detachment were compared between the two groups.
Transplantation of mesenchymal stem cells
Twenty-four hours after induction of retinal injury, bone marrow-derived MSCs were transplanted into the four rats of the experimental group. After inhalation anesthesia with diethyl ether, 4 µL of cell suspension containing 1 × 106 cells was systemically administered via tail vein in the experimental group. In the four rats of the control group, 4 µL of 0.1 M PBS was injected.
Histopatholgical analysis
At five and seven weeks after MSCs injection, two eyes from the experimental group and two eyes from the control group were enucleated for tissue preparation. Xylazine hydrochloride (4 mg/kg) and ketamine hydrochloride (10 mg/kg) were intramuscularly injected for anesthesia. The thoracic cavity of each animal was opened, then 500 mL of 4% paraformaldehyde (Merck, Darmstadt, Germany) was perfused through the left ventricle. The eyeballs were enucleated and then placed in 4% paraformaldehyde in 0.1 M PBS overnight. To prevent tissue injury in making a frozen section, the eyes were placed in 10%- and 20%-sucrose solutions for one hour, respectively. Following this, they were placed in a 30% sucrose solution for another 12 hours. The horizontal plane of the eyeball was positioned parallel to the cutting plane. A tissue section 4 µm in thickness was prepared. Changes to the injured retinal lesions were observed using a light microscope (E400; Nikon, Kanagawa, Japan) after Wright stain. To evaluate the homing capacity of transplanted MSCs to the retina, the presence of GFP expressing cells was detected using a confocal microscope (ECLIPSE TE300, Nikon).
Results
Confirmation of GFP-transduced MSCs
The expression of GFP in these cells was confirmed by confocal microscopy. After adhesion to the plastic culture flask, MSCs exhibited the typical spindle-like morphology and were arranged in a foray fashion. Adhesion to the culture flask also served as a criterion to distinguish MSCs from free floating hematopoietic cells.
Observation of fundus findings
The injury sites were studied at one day after retinal injury and at three weeks and five weeks after MSCs transplantation using fundus photography (Fig. 1). For the fundoscopic examination at day one after treatment with the Nd:YAG laser, hemorrhagic retinal elevations were observed at the injury site in all eight eyes (Fig. 1A and 1D). Three weeks after transplantation, the extent of subretinal and vitreous hemorrhages decreased in all four eyes of the experimental group and proliferative lesions were not observed in all four fundi of the experimental group (Fig. 1B). During that time, the hemorrhagic components in the eyes of the control group seemed to diminish in size. However, definitive elevations in the neurosensory retina were detected around the thick hemorrhage site (Fig. 1E). In the experimental group, the hemorrhagic components were much decreased and the ruptured retinal sites could not be detected in the color fundus photograph at five weeks after transplantation (Fig. 1C). However, the retinal detachment that developed in the eyes of the control group was more progressed with the proliferative changes and the hemorrhagic components remained at the same time point (Fig. 1F).
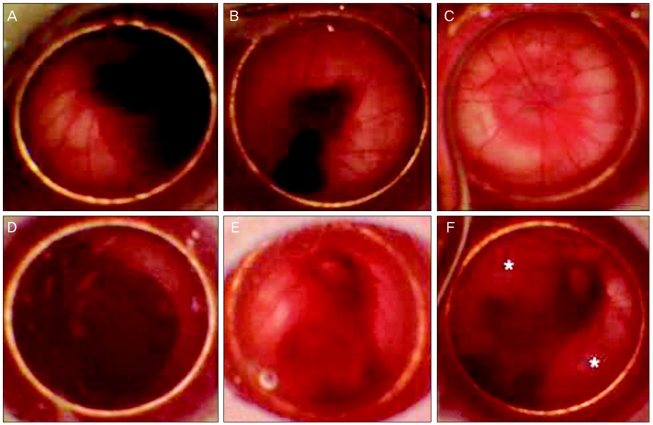
Fundus photographs showing time-dependent fundus changes followed by laser induced retinal trauma in the experimental group (A-C) and control group (D-F). (A) One day after retinotomy, large diffuse subretinal hemorrhages were observed. (B) Three weeks after transplantation, diffuse subretinal hemorrhages subsided compared with previous findings. (C) Definite hemorrhagic lesions were not detected at five weeks after transplantation. (D) One day after retinotomy, thick vitreous hemorrhages with retinal detachments were seen. (E) At three weeks, retinal detachments remained and hemorrhages were observed. (F) At five weeks, retinal detachment (*) remained in the control group.
Evaluation of homing capacity and wound healing processes
Histological analysis with Wright staining and GFP expressing cell detection using confocal microscopy were performed on animals at five and seven weeks after MSCs transplantation. At five weeks after treatment with intravenous injection of 1 × 106 MSCs, the laser-induced retinotomy site was partially closed with uncertain types of cell cluster. Round-shaped, dark-pigmented cells were observed at the basal area of the full thickness damaged site. Cellular components of the injured site were certainly distinguished as compared with surrounding healthy retinal tissues. Only two or three layered unidentified cells were detected in the damaged site and there were no properly arranged neuronal networks between the various types of neuronal cells (Fig. 2A and 2B). To investigate whether bone marrow-derived MSCs were incorporated into the damaged chorioretinal tissues, we searched the serially sectioned slides around the damaged site. GFP expressing bone marrow-derived cells were detected only in the laser-induced injury site (Fig. 3A-3C). The placements of GFP expressing cells corresponded to those of round dark-pigmented cells observed in Fig. 2B. These findings, therefore, suggest that transplanted adult bone marrow-derived MSCs can contribute to the repair of injured chorioretinal tissues in the retinal trauma model. The round pigmented cells, as shown in Fig. 2B, were not detected in the injured retina at seven weeks post transplantation, but the laser-induced retinal break at the damaged site was much recovered with unidentified numerous cellular populations (Fig. 2C and 2D). Compared to the injured site at five weeks, the thickness of the cellular component increased and the injured site was much stabilized at seven weeks. Although normal retinal structure could not be identified in the lesion site, retinal detachment was not developed and the number of GFP expressing cells significantly increased in the laser-induced retinotomy site at seven weeks after transplantation (Fig. 3D-3F).
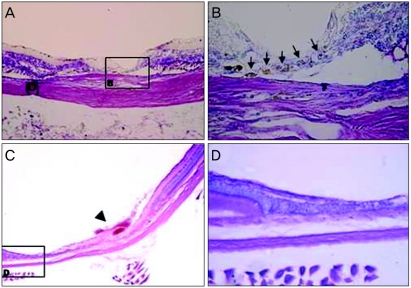
Light microscopy findings for the laser-induced retinotomy site at five and seven weeks after mesenchymal stem cells transplantation in the experimental group. (A) As observed, damaged neurosensory retina lacked continuity (Wright staining, ×100). (B) Note that the lineal cluster of round and dark pigmented cells (arrow) made a retinal hole that was closed partially at the base of the iatrogenic hole area (Wright staining, ×400). (C) Broad healed retinal tissue was seen. A fibrotic horn-like structure (arrow head) seemed to indicate the precise location of laser induced physical trauma. (D) After the wound healing process, the lesion was completely covered with unidentified cells without retinal detachment, however this was different than those of normal neurosensory retina. Only intermingled cell clusters without systemically layered neuronal structures were observed. The outer segment of the photoreceptor cell disappeared abruptly in the transitional zone between healed and undamaged retina.

Confocal microscope images of the laser-induced retinotomy site in a continuous section at five and seven weeks after mesenchymal stem cells (MSCs) transplantation (A-C, ×200; D, ×100; E, ×200; F, ×400). (A) Green fluorescence protein (GFP) positive cells were identified in the damaged chorioretinal tissue. (B) Propidium iodide was used for nuclear counterstaining. (C) Merged image of A and B, note that GFP positive cells were perfectly identical to the round, pigmented cells seen in Fig. 2B. (D-F) GFP positive cells originated from MSCs were widely distributed in the healed retina. The number of GFP positive cells increased compared to the images at five weeks after transplantation.
Discussion
Striking features common to the retinas of both amphibians and fish include persistent neurogenesis throughout life and injury-induced neural regeneration. The death of retinal neurons stimulates regenerative neurogenesis. In these animals, the injured retinas provide an environment that is permissive for the proliferation and differentiation of neuronal progenitors [21]. In contrast, the regeneration of neuroretinal cells in mammals is extremely limited. Therefore, recently, various kinds of attempts have been made to manage degenerative retinal diseases with embryonic stem cells, bone marrow-derived stem cells, and some kinds of neural progenitor cells [22,23]. However, these approaches with embryonic stem cells remain highly controversial and contain huge obstacles such as rejection and potential tumorigenesis. The harvest and preparation of neural progenitor cells for autologous stem cell therapies is complicated by the deep location of these cells in the brain in areas such as the hippocampus [24,25]. Therefore, MSCs seem to have significant advantages as sources of cells for cell replacement therapy.
To date, tissue and cells have been transplanted into the subretinal space and various types of precursor cells have been injected into the vitreous cavity [9,15-17,24]. In the present study, the authors observed that GFP expressing MSCs were identified at the injured retina. These findings suggest that MSCs can target and be incorporated into the neuroretinal layer, although they were systematically injected via a different method from the previously mentioned literature. Nishida et al. [17] attempted to transplant neural stem cells derived from the hippocampus by injection into the vitreous cavity. These authors noted that the transplanted stem cells did not migrate to normal retinal tissue but rather to retina tissue in which mechanical injury was induced using a fine needle. Minamino et al. [24] reported that bone marrow stem cells injected into the vitreous cavity without pre-treatment did not migrate into the retina but rather the stem cells migrated effectively following the laser photocoagulation. Also in the present study, GFP expressing cells were not identified in the normal retinal tissue at weeks five and seven. Instead, these cells were distributed only in the junction between the injured and normal retina. Pre-treatment of the normal retina was needed to incorporate the implanted cells effectively, a result which is identical to previous reports. Our results suggest that the Nd:YAG laser induced retinotomy model might be another experimental model for the transplantation of stem cells.
Damaged retina triggers cellular injury and inflammatory responses and also releases various types of growth factors and inflammatory cytokines. Basic fibroblast growth factor plays a key role in the proliferation of MSCs and vascular endothelial growth factor regulates the survival and reproliferation of myeloblasts. Stromal-derived growth factor-1 is known to assist in the migration of bone marrow cells to the damaged site [26-29]. We speculate that in the present study these growth factors might have stimulated the transplanted MSCs to target the retina and incorporate effectively into the injured retinal lesion.
The retinal lesions injured with the Nd:YAG laser demonstrated a contrary process. The retinotomy sites were completely closed and the accompanying hemorrhage lesion was resolved in the MSCs transplanted eye at seven weeks after transplantation. On the other hand, in the control group the retinotomies developed into retinal detachment with huge subretinal hemorrhages. In this respect the results of our study support the possible implementation of a cell-based strategy using MSCs for potential wound healing modulation in physically injured retina. To date, experimental studies have not presented definite answers regarding the role of implanted bone marrow MSCs and related mechanisms. However, several hypotheses have been proposed: 1) transplanted stem cells play the same role as true MSCs, replace the stem cell group in the specific tissue, and form the new connective tissue. Then, growth is achieved in a similar way as in normal tissue. 2) MSCs or their progenitor cells can be transformed and differentiated under specific microenvironments. Therefore, they can form endodermal or ectodermal tissue as well as mesenchymal tissue. 3) Although seen in particular situations, heterozygotes can be formed between the transplanted bone marrow MSCs and the specific cells of the recipient tissue. 4) The transplanted MSCs release paracrine factors and thereby induce the proliferation of specific cells in the transplanted tissue or contribute to making the microenvironment more appropriate for the repair of injury by maximally inhibiting the tissue injury [1].
In the present study, we did not perform experiments to evaluate the differentiation patterns of the incorporated MSCs. Exactly what cells types the transplanted MSCs differentiated into is therefore unclear. Further studies are needed in this series. In addition, a molecular biological approach will be made in order to elucidate the mechanism for the cell migration and wound healing.
In conclusion, although there were some limitations regarding the exact wound healing mechanism and differentiation of transplanted MSCs, it is clear that systemically administered MSCs have the capacity to specifically target injured rat retina and play important roles in the wound healing process. Together with findings from studies using other routes for stem cell incorporation or other cell based treatments, the results from our experiment involving MSCs indicate that wound healing effects were partially achieved and point to an additional direction of the route of stem cell administration.
Notes
No potential conflict of interest relevant to this article was reported.