Short Term Effects of Topical Cyclosporine and Viscoelastic on the Ocular Surfaces in Patients with Dry Eye
Article information
Abstract
Purpose
To compare the short term effects of topical 0.05% cyclosporine (CsA) and a mixture of 0.08% chondroitin sulfate and 0.06% sodium hyaluronate (CS-HA) on dry eye ocular surfaces.
Methods
36 patients with moderate to severe dry eye (5 mm/5 min or less with Schirmer's test or tear break up time (BUT) less than 6 seconds), were treated with topical application of CS-HA on one eye and CsA on the other 4 times a day for 6-8 weeks. BUT, Schirmer's test without anesthesia, and conjunctival impression cytology (CIC; goblet cell density, nucleus to cytoplasmic ratio, and epithelial cell morphology) were evaluated and compared between eyes before and after treatment (repeated measurement of ANOVA).
Results
After treatment, BUT and tear wettings were significantly prolonged in each group. Topical CsA treated eyes had greater increase in BUT (p=0.026); there was no significant difference in tear wetting (p=0.132). While the 3 parameters of CIC improved in both groups, goblet cell density was significantly higher in eyes treated with CsA (p=0.033).
Conclusions
While both CS-HA and 0.05% CsA eyedrops improve ocular surfaces, topical CsA may have a better effect on enhancing tear film stability and goblet cell density.
Dry eye is a tear film disorder caused by tear deficiency or excessive evaporation, and is associated with ocular discomfort, dryness, scratchiness, burning, soreness, and grittiness.1 Objective signs of ocular surface damage include tear film instability and tear hyperosmolarity.2
In recent years, there has been growing evidence that inflammation and apoptosis of the ocular surface has a key role in the development of dry eye.3,4 In addition, inflammatory cytokines such as interleukin (IL)-1 α, IL-8, and tumor necrosis factor-α, and immune-activation marker expression, such as HLA-DR, intracellular adhesion molecule-1, and CD11 a, are increased in dry eyes.4,5 This chronic inflammatory environment is partly responsible for the characteristic conjunctival epithelium pathologic alterations, such as squamous metaplasia and goblet cell loss.6
Therefore, recent trends in KCS treatment have been aimed at improving ocular surfaces by suppressing apoptosis and conjunctival epithelium and goblet cell inflammation.7
Topical cyclosporine A (CsA) has been reported to decrease immune- and apoptosis-related markers,8 improve aqueous tear production,9 increase conjunctival goblet cell density,4 and have a therapeutic effect on various ocular surface diseases.10
Similarly, topical sodium hyaluronate (HA) has been reported to suppress apoptosis-related and inflammatory markers (Fas, Apo 2.7, HLA-DR and CD 40), upregulate protective markers (MUC5AC and CD63),8 and increase goblet cell density and aqueous tear production.11,12 Chondroitin sulfate (CS), used as a lubricant,12 has also been reported to have anti-inflammatory effects.13
Although topical CsA and CS-HA have anti-inflammatory and apoptosis-suppressing effects, no study has compared these two preparations. The purpose of this study was to investigate and compare the short term effects of topical CS-HA and CsA on the ocular surfaces of dry eye patients.
Materials and Methods
Patient characteristics
Informed consent was obtained from all subjects, and the study was approved by our institutional review board. Forty-seven consecutive patients with bilateral moderate to severe dry eye were enrolled prospectively. The female-to-male ratio was 38:9. Age distribution was 49.31±11.38 years. Moderate to severe dry eye disease was defined by: (1) Schirmer's test reading without anesthesia of less than or equal to 5 mm / 5 min14; or (2) 3 tear film BUT all less than 6 seconds.15 Topical 0.08% chondroitin sulfate (CS) mixed with 0.06% sodium hyaluronate (HA) was applied on one eye and 0.05% cyclosporine (CsA) on the other eye four times a day for 6-8 weeks. There was at least a 2-week washout phase before starting treatment to eliminate previous effects of artificial tears. The mean application was 48.37±9.61 days. In this period, compliance was evaluated by telephone interview and was found to be similar between the two eyedrops. Poor compliance was found in 11 participants, who were excluded from the study.
We excluded patients who reported topical or systemic CsA or ophthalmic steroids use within the previous 90 days or 3 weeks, respectively. Other reasons for exclusion included previous punctual plug, contact lens wear, use of other topical treatments, active ocular infection, severe blepharitis, recurrent herpes keratitis within the previous 6 months, anterior segment surgery or trauma within the previous 12 months, punctuate epithelial erosions involving more than one third of the cornea, other ocular surface diseases, or non-KCS inflammation, including atopic keratoconjunctivitis.
Eyedrop Preparation
CS-HA and CsA eyedrops were prepared at Seoul National University Hospital pharmacy under sterile conditions. CS-HA eyedrops were made by diluting 0.75 ml Viscoat® (a 1:3 mixture of 4% CS and 3% HA; Alcon Surgical, Inc., Fort Worth, TX) in 35 ml normal saline, producing topical viscoelastics containing 0.08% CS and 0.06% HA (CS-HA eyedrops). CsA eye drops were made by mixing Neoral® Soft Gelatin Capsules (Novartis, Japan) containing 100 mg of micro-emulsified cyclosporine (1 ml) with 19 ml balanced salt solution (BSS®, Alcon Surgical, Inc., Fort Worth, TX), producing topical 0.5% CsA.; 9 ml balanced salt solution was added to 1 ml of 0.5% CsA to make 0.05% eyedrops. Preparations were maintained regularly to avoid contamination and loss of efficacy.
Monitoring effects of topical eyedrops
Tear film break-up time (BUT), Schirmer test without anesthesia, and conjunctival impression cytology (CIC) were performed before and after scheduled instillation (mean interval: 48.37±9.61 days). The examiner was blind to eyedrop type. Tear film BUT was measured three times to calculate a mean value. Schirmer's test without anesthesia was performed using standard strips (Alcon, Fort Worth, Texas, USA) in the lower conjunctival sac for 5 minutes. CIC was performed as follows. After topical anesthesia with 0.5% proparacaine hydrochlororide (Alcaine®, Alcon- Couvreur, Puurs, Belgium), MF Millipore membrane filters with 0.22 µm pores were applied to the temporal bulbar conjunctiva. To guarantee the correct staining surface, the membrane filter was cut into 5×5 mm asymmetrical pieces with a pointed tip in one corner. Using an ophthalmodynanometer, 60 g of gentle pressure was applied to the filter for 5 seconds; the membrane was fixed in 95% ethanol, stained with periodic acid-Schiff, dehydrated in ethanol (70, 80, 90, 100% in sequence) and xylene (80, 90, 100% in sequence), and then coverslipped. Using 400 magnification optical microscopy goblet cell densities (GCD), nucleus to cytoplasm ratios (NCR) and epithelial cell morphology (ECM) were evaluated. GCD was averaged for every 100 epithelial cells in five high-power fields (HPF, ×400 magnification) in each eye. NCR and ECM, graded according to Saini et al.16 were defined as the average value for five HPF in each eye.
Statistical analysis
Statistical analysis was performed using SPSS for Windows version 11.5 (SPSS, Chicago, IL, USA). To compare therapeutic effects between eyedrops, ANOVA was used. P<0.05 was considered significant in all statistical testing.
Results
Tear film BUT
Before eyedrop application, there was no significant difference of tear film BUT between each group of eyes (4.19±0.86 seconds on CS-HA-treated eyes and 4.08±1.01 seconds on CsA-treated eyes, p=0.623). After scheduled applications, there was significant prolongation of tear film BUT in both groups (4.39±1.12 sec on CS-HA-treated eyes and 5.08±0.77 on CsA-treated eyes), (p=0.041 and 0.029, respectively). BUT increase was more prominent in the CsA group than in the CS-HS group (p=0.026, Fig. 1).

Comparing tear film BUT between eyes before and after topical application of topical 0.077% chondroitin sulfate with 0.06% hyaluronate (on right eyes) and 0.05% cyclosporine (on left eyes). Before treatment, there was no significant difference between eyes. After applications, tear film BUT increased significantly in both eyes but was more profound in left eyes.
Schirmer's test
There was no significant difference of aqueous tear production between the two groups before treatment (5.72±1.72 seconds on topical CS-HA treated eyes and 6.17±1.56 seconds on topical CsA treated eyes) (p=0.234). Tear wetting increased significantly in both groups compared with pretreatment levels (6.47±1.68 seconds on topical CS-HA treated eyes and 7.17±1.56 seconds on topical 0.05% CsA treated eyes), (p=0.024 and 0.011, respectively). Although the amount of tear wetting tended to be greater in the 0.05% CsA group, there was no statistically significant difference between the two groups (p=0.132) (Fig. 2).
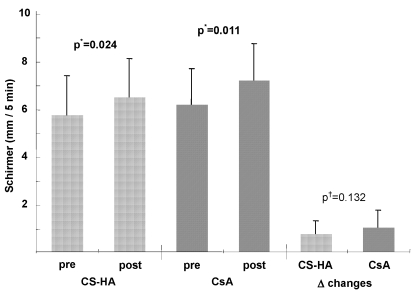
Comparing aqueous tear production (Schirmer's test) between eyes before and after topical application of 0.077% chondroitin sulfate with 0.06% hyaluronate (on right eyes) and 0.05% cyclosporine (on left eyes). After treatment with the assigned eyedrop, aqueous production increased significantly in both eyes. The amount of increase in both eyes was similar.
Conjunctival impression cytology
Before treatment, mean GCD, NCR, and ECM were 5.36±1.02, 3.27±0.97, and 4.25±0.61 in topical CS-HA treated eyes and 5.47±1.71, 3.53±0.61, and 4.59±0.78 in topical CsA treated eyes, respectively with no significant difference between the two groups (p=0.795, 0.333, and 0.688, respectively). After scheduled treatment with topical CS-HA, there was significant improvement of GCD, NCR, and ECM (p=0.047, 0.035, and 0.018, respectively) (Fig. 3, 4, 5). Similarly, there was marked improvement of GCD, NCR, and ECM in topical CsA treated eyes (p=0.012, 0.025, and 0.027, respectively) (Fig. 3, 4, 5). GCDs increased more prominently in topical CsA treated eyes than in topical CS-HA treated eyes (p=0.033), while NCR and ECM showed no significant difference (p=0.721 and 0.439, respectively) (Fig. 3, 4, 5).
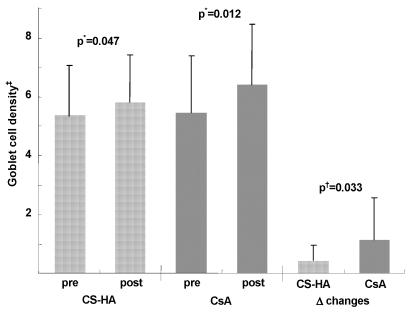
Before treatment with topical eyedrops, goblet cell densities (GCD) were similar between eyes. After assigned treatments (topical chondroitin sulfate with sodium hyaluronate (CS-HA) for right eyes and topical cyclosporine (CsA) for left eyes), there was a significant GCD increase in both eyes. However, GCD increase was greater in left eyes.
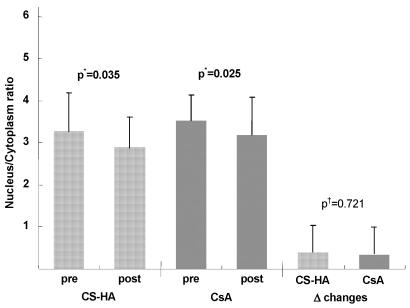
Before topical eyedrops, nucleus to cytoplasmic ratios (NCR) of conjunctival impression cytology were similar between the two groups. After assigned treatments (topical chondroitin sulfate with sodium hyaluronate (CS-HA) for right eyes and topical cyclosporine (CsA) for left eyes), there was significant NCR improvement in both eyes. There was no significant differences among treatment groups.
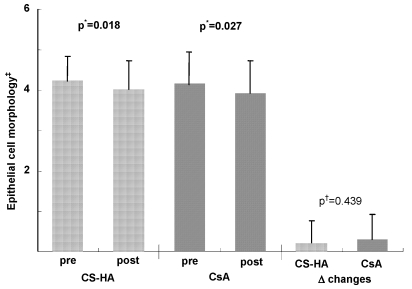
Before treatments, epithelial morphologies (ECM) of conjunctival epithelium in conjunctival impression cytology were similar between eyes. While there was significant ECM improvement in both eyes after treatments (topical chondroitin sulfate with sodium hyaluronate (CS-HA) for right eyes and topical cyclosporine (CsA) for left eyes), the degree of change in both eyes was similar.
Discussion
Conjunctival goblet cell densities are sensitive indicators of ocular surface disease. In dry eyes, the first evidence of surface injury is the manifestation of decreased conjunctival goblet cells.17 The results of this study suggest that topical CS-HA and 0.05% CsA stabilize tear film instability, increase aqueous tear production, and also improve ocular surfaces; these results are consistent with previous studies.18
Kunert et al. reported that topical 0.05% CsA increased conjunctival goblet cell density and decreased epithelial turnover after 6 months of application.4 Strong et al. also demonstrated that topical CsA significantly reduced conjunctival epithelial apoptosis and protected against goblet cell loss in experimental murine keratoconjunctivitis sicca.19 However, the novelty of this study is that 0.05% CsA eyedrops increased conjunctival goblet cell density and tear film stability more effectively than CS-HA. This result can be explained as follows. Ocular surface mucin, consisting predominantly of conjunctival goblet cell secretions, adheres to conjunctival and corneal epithelial cells glycocalyces and enhances corneal wettability by serving as an interface between the hydrophobic corneal epithelium and aqueous tear fluid. We believe that mucin increases with increased goblet cell density, which was enhanced by topical cyclosporine. This explanation is also supported by Moore et al.; they found that topical cyclosporine restored in vivo dog conjunctival mucin stores to control levels over a 4-week period.20
Commercial topical cyclosporine was not available in our country at the start of the study, and was difficult to prepare due to hydrophobicity (log P=3.0) and low aqueous solubility (6.6 ug/ml).21,22
We prepared 0.05% topical CsA by mixing CsA capsule contents (Neoral® Soft Gelatin Capsule, Novartis) with a balanced salt solution. In this capsule, CsA is dissolved in ethanol with corn oil-mono-di-triglycerides, propylene glycol (an enhancer of tissue permeation by supersaturation systems, based on increased thermodynamic activity), and polyoxyl 40 hydrogenated castor oil (a pharmaceutical aid (nonionic surfactant) used to improve solubilization). Propylene glycol is tissue permeation enhancer by supersaturation systems. Kuwano et al. demonstrated that CsA solubility into aqueous formulations containing polyoxyl 40 hydrogenated castor oil.23 In an aqueous medium with nonionic surfactant, micelles are formed, allowing CsA to dissolve in them. As micelle size is smaller than emulsion droplets, with a mean diameter of 200 nm, they have a larger surface area, enabling more drug binding. Therefore, more CsA can be released for ocular surface delivery. This may explain the early therapeutic effects of topical CsA. The main downside to our preparation was a stinging sensation in patients with moderate epithelial erosion, possibly caused by sub-appropriate pH. Therefore, we limited 0.05% CsA application to patients without epithelial defect or erosions involving more than one third of the cornea.
Topical eyedrops containing 0.08% CS and 0.06% HA were prepared by diluting commercial viscoelastic material in normal saline. The diluting concentration was determined from previous reports.12,24
Topical application of HA has been shown to result in both subjective and objective improvements.13,25 Several mechanisms have been suggested. HA is a natural biopolymer and its concentration increases in response to ocular damage and corneal wound healing.26 In vitro HA promotes cell migration and stabilizes the ocular surface epithelial barrier,27,28 suggesting intrinsic wound healing properties. Binding of HA to CD44, a corneal and conjunctival cell receptor,29 may promote cell adhesion and motility via cytoskeletal protein action,30 stimulate cell proliferation through a kinase cascade mechanism,31 and enhance rabbit corneal epithelial cell growth in vitro.32 Interestingly, it has been reported that CD44 is expressed in 30-50% of conjunctival epithelial cells in patients with moderate dry eye, and that HA is associated with decreased expression.8 Apoptosis-related and inflammatory markers (Fas, Apo 2.7, HLA-DR, and CD40) tend to decrease and potentially protective markers (MUC5AC and CD63) tend to increase with HA application.8
HA also increases precorneal tear film stability, which protects the ocular surface from environmental agents11 and has water retentive properties improving wettability.33 Therefore, HA may contribute to a favorable microenvironment during blinking and ocular movements.
In our study, squamous metaplasia on CIC improved after treatment with CS-HA eyedrops for 48.37±9.61 days. The recovery time of patients in our study seems to be shorter than that of previous reports of 8 weeks or more.34 This discrepancy can be explained by the synergistic anti-inflammatory effect of CS. CS is known to downregulate gene expression and synthesis of nitric oxide synthase and prostaglandin E2 production,35 components of the inflammatory cascade, and induce IL-1β, an inducing factor for lacrimal gland cell death.35 As IL-1β and matrix metalloproteinase-9, a physiological activator of IL-1β, are significantly elevated in tear fluid and conjunctival epithelium of dry eyes,36,37 the suppressive effects of CS can enhance HA effects to improve ocular surfaces.
Given the immediate symptomatic improvements and synergistic anti-inflammatory effects of topical CS-HA, it can be recommended that topical CS-HA be simultaneously administered with CsA, which may require longer treatment to suppress apoptosis. Additionally, as HA and topical CsA have similar effects via the different aforementioned mechanisms, it is possible that HA might act synergistically with topical CsA; further investigation would be helpful.
CS-HA and CsA eyedrops were mixed with normal saline and a balanced salt solution, respectively, which may have had effects on ocular surfaces or tear production. Because a vehicle-treated control group was not included, this perspective cannot be evaluated. Another limitation is the inability to demonstrate the effects of CS-HA or CsA on each patient's other eye, which may have been mediated by the neural loop of the ocular surface lacrimal functional unit.
Conclusion
This study suggests that, while topical applications of CS-HA and CsA are effective for dry eye, topical CsA may have a greater effect on the improvement of conjunctival GCD, which can enhance tear film stability, compared with topical CS-HA.
Notes
This study was supported by the Korea Health 21 R&D Project. Authors have no conflicts of interest. Authors have no financial interests in the materials or methods mentioned.